Similarly, there are set of five d orbitals ( l = 2).
In case of s and d orbitals the wave functions have the same sign at the same distance in opposite directions from the center of symmetry. These orbitals are thus centrosymmetric and are said to be gerade . It means even and denoted as g . While, the wave functions for p orbitals have opposite signs on either side from the centre of symmetry. Therefore, they are known as non-centrosymmetric or ungerade orbitals. They are denoted as u .
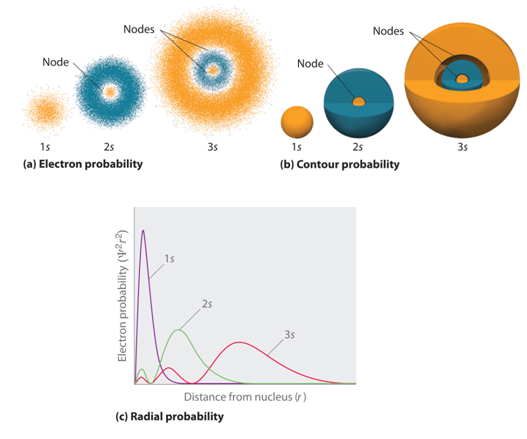
Angular probability function :
The probability of finding an electron simaltanously at a distance r and in a given direction Φ , θ is
Ψ 2 r, Φ, θ
![]()
The probability of finding an electron in a given direction is then A 2 ( Φ , θ ).
s orbital is spherical so it remains unchanged on squring, while, for p and d orbitals sign factor vanished and lobes becomes more elongated.
Difference between orbits and orbitals:
Orbit represent discreate trajectories or electrons around the positively charged nucleus. The position, momentum, and consequently, energy of an electron can be expressed precisely by using laws of newtonian mechanice assuming electron as particle.
The idea of orbital was introduced by the wavemechanical atom model. The extranuclear part has been considered as wave and follows the fundamental Schodinger wave equation. The orbital corresponds to the probability of finding an electron at a certain distance and certain direction. Hence, each orbital has definite energy, shape and described in terms of quantum numbers.