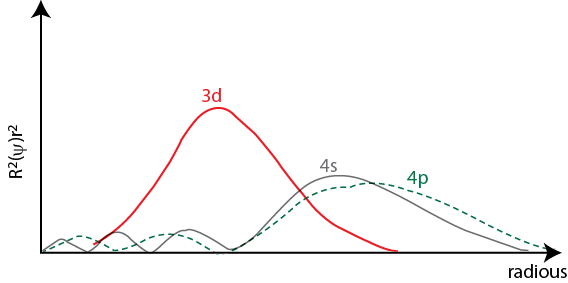
Figure 1.8.
Significance:
- The probability function is always positive as it involves the square of R .
- At r = 0, the probability of finding electron is zero. This shows no existence of electron at the nucleus.
- R is a function of n and l . Therefore, with increase in value of either n or l , the distance of finding maximum electron density form the nucleus will also increase.
- At a large value of r, R approaches to zero. Therefore, the probability of finding electron will approach to zero.
- Most probable distance increases as the principle quantum number ( n ) increases.
- Most probable distance decreases slightly as the subsidiary quantum number ( l ) increases.
Penetration:
Penetration of orbitals means their distribution into inner electronic core. For example, the radial density of 2s orbital spread into the curve for 1s orbital. Similarly, 3s orbital is spread into 1s, and 2s orbital. Because of this spreading electrons in 2 s orbital or in 3 s orbital will not be fully screened by the inner 1s electrons from the nucleus. The extent of penetration decreases from s to f orbitals.
s > p > d > f
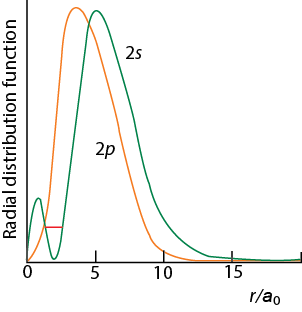 |
Figure 1.9. Penetration decreases from s to p as radial distribution close to nucleus for s is more compared to p . |