The dependence of ψ on r , θ , and Φ can not be shown directly with equation (xxi). Because, it would require a four dimensional graph. However, the equation in this form can be express as follows,
(xxii) |
- R (r) is a function that depends on the distance from the nucleus. It depends on the quantum numbers n and l .
- Θ (θ) is a function of θ and depends on the quantum numbers l and m .
- Φ (Φ) is a function of Φ and depends on the quantum numbers m .
Therefore, equation (xxii) can be express as,
![]()
This splits wave function into two parts which can be solved separately,
- R ( r ) is a radial function that depends on the quantum numbers n and l .
- A ml is the total angular wave function that depends on the quantum numbers m and l .
Radial part of wave functions, R :
The radial function R has no meaning. R 2 gives the probability of finding the electron in a small volume d v near the point at which R is measured.
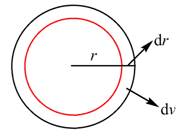
Figure 1.6. Showing volume difference
For a given value of r the total volume will be,
![]()
We may consider that an atom is composed of thin layers of thickness d r . The volume d v for between r and r +d r will be then (Figure 1F),
![]()
The probability of finding the electron in that volume will be,
![]()
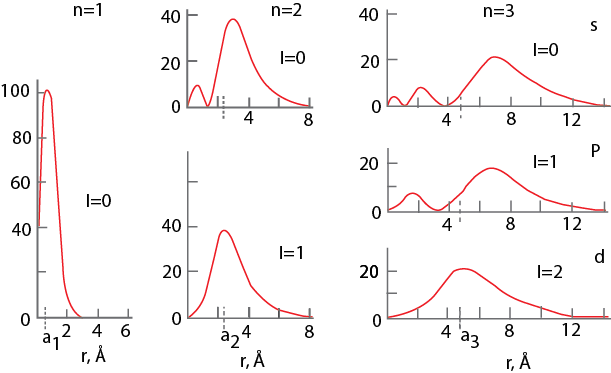
Figure 1.7. Radial probability functions for n = 1, 2,3 for the hydrogen atom. The radial density is along y axis.