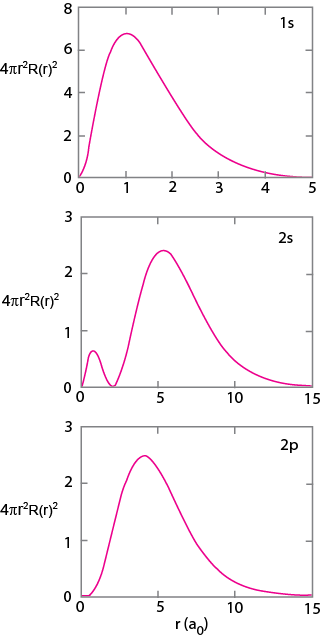
 |
Figure 1.10. Radial distribution of 1s, 2s, and 3s orbital. |
Angular part of wave functions:
The angular parts of the wave function depend only on direction ( Φ and Θ ) are independent of the value of principle quantum number ( n ) and the distance from the radius ( r ). Thus, A 2 is the probability of finding an electron at a given direction Φ and Θ at any distance from the nucleus to infinity. They depend primarily on the values of l . For l = 0, i.e. s orbitals, the angular wave function is constant and independent of the angles θ and Φ . Therefore, this function will provide a sphere.
Angular functions for some orbitals,
![]()
Here, z or z 2 refer the orientations of the orbitals along z axis. The angular wave function of the p z orbital corresponds to two spheres whose center lies on z axis. The spheres touch each other tangentially at the origin. Since, cos θ changes sign with θ , the wave function will have two different sign in two different lobes.
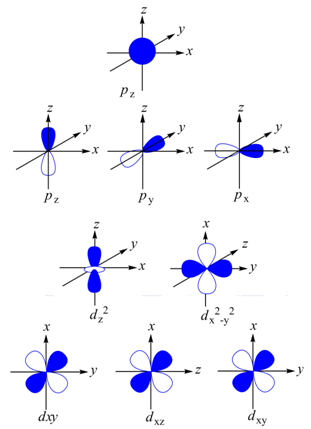
Figure 1.11. The angular part of the wave function A ( θ ,. Φ ) for the 1 s , 2 p, and 3 d orbitals for a hydrogen atom.