The hydrogen atom:
Schrödinger equation for hydrogen atom:
Schrödinger equation can be solved completely for hydrogen atoms as well as hydrogen type atoms, like, He1+ , Li2+ ( Z = 1). For the other atoms only approximate solution can be achieved.
For most calculations, it is simpler to solve the wave equation if the Cartesian coordinates x , y , and z are converted to polar coordinates, r , θ , and Φ .
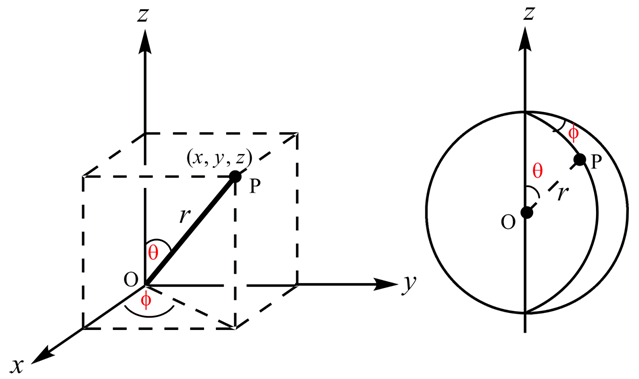
Figure 1.5. Cartesian and polar coordinates.
It can be seen from Figure 1E that two sets of coordinates are related to each other by the following relation,
z = r cos θ
y = r sin θ sin Φ
x = r sin θ cos Φ
The Schrödinger equation is written as,
(xix) |
Where,
![]()
Changing to polar coordinates, ∇2Ψ becomes,
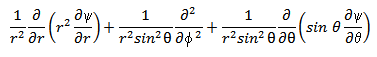
Now we can write equation (xix) as,
(xx) |
(xxi) (Potential energy ( E k ) = V , see equation vii) |