Staged Combustion
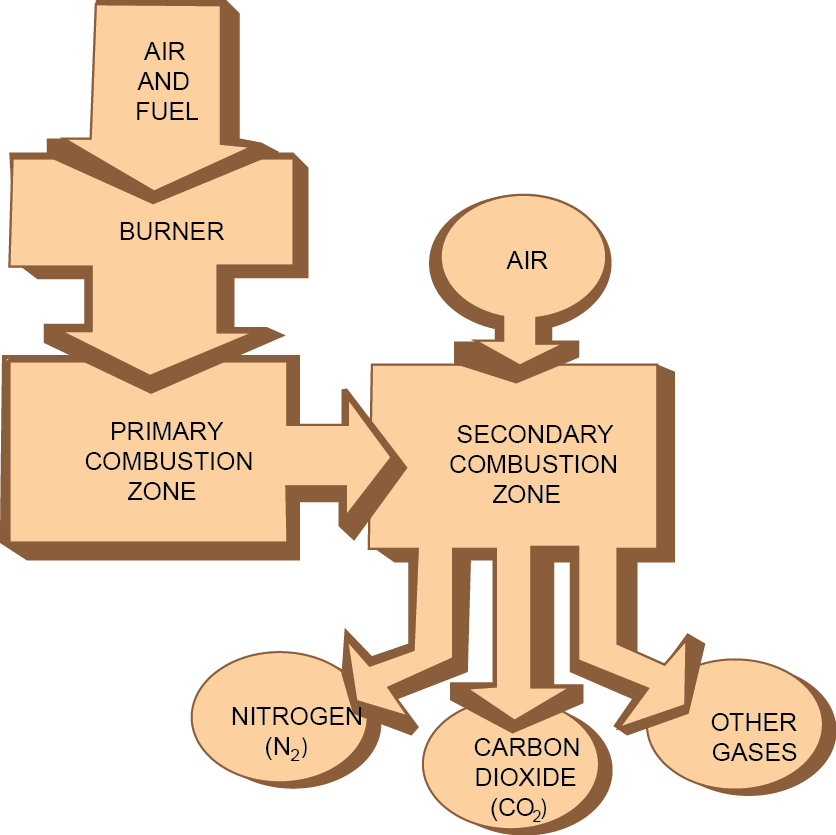
PRINCIPLE
- Initially,less air is supplied to bring about incomplete combustion
- Nitrogen is not oxidized. Carbon particles and CO are released.
- In the second stage, more air is supplied to complete the combustion of carbon and carbon monoxide.
30% to 50% reductions in NOx emissions are achieved.
Selective noncatalytic reduction systems make use of the tendency of certain compounds, ammonia ((NH3) and urea (H2NCONH2) in particular, to react with and reduce oxides of nitrogen:
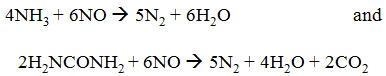
When either ammonia or urea is injected into flue gases at temperatures of about 870°C–1,100°C for ammonia and 900°C–1,150°C for urea, NOx removal can reach as high as 50 percent.
Selective catalytic reduction operates on the same principle as SNCR, using ammonia or urea as the working gas along with a catalyst.
A number of catalysts have been used, the most effective of which appear to be oxides of molybdenum, titanium, tungsten, and vanadium as well as zeolites, which are naturally occurring alumina silicates.