The post-combustion systems used at power generating plants and factories are somewhat different.
These systems remove nitrogen oxides from the waste gases (flue gases) processes using classified as selective noncatalytic reduction (SNCR) and selective catalytic reduction (SCR).
Oxides of nitrogen are also removed by some systems used to extract sulfur dioxide from flue gases.
Selective Catalytic Reduction (SCR)
- In this process, the nitrogen oxides in the flue gases are reduced to nitrogen
- During this process, only the NOx species are reduced
- NH3 is used as a reducing gas
- The catalyst is a combination of titanium and vanadium oxides. The reactions are given below :
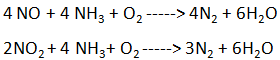
- Selective catalytic reduction catalyst is best at around 300 too 400 oC.
- Typical efficiencies are around 80 %
Selective Non-catalytic Reduction (SNR)
- At higher temperatures (900-1000oC), NH3 will reduce NOx to nitrogen without a catalyst.
- At NH3: NOx molar ratios 1:1 to 2:1, about 40-60%reduction is obtained.
- SNR is cheaper than SCR in terms of operation cost and capital cost.
- Tight temperature controls are needed. At lower temperatures, un-reacted ammonia is emitted. At higher temperatures ammonia is oxidized to NO.
Selective noncatalytic reduction systems make use of the tendency of certain compounds, ammonia (NH3) and urea (H2NCONH2) in particular, to react with and reduce oxides of nitrogen:
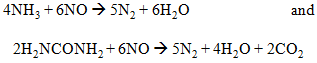
When either ammonia or urea is injected into flue gases at temperatures of about 870°C–1,100°C for ammonia and 900°C–1,150°C for urea, NOx removal can reach as high as 50 percent.
Selective catalytic reduction operates on the same principle as SNCR, using ammonia or urea as the working gas along with a catalyst.
A number of catalysts have been used, the most effective of which appear to be oxides of molybdenum, titanium, tungsten, and vanadium as well as zeolites, which are naturally occurring alumina silicates.
Electron Beam Radiation
- This treatment process is under development, and is not widely used. Work is underway to determine the feasibility of electron beam radiation for neutralizing hazardous wastes and air toxics.
- Irradiation of flue gases containing NOx or SOx produce nitrate and sulfate ions.
- The addition of water and ammonia produces NH4 NO3, and (NH4)2SO4
- The solids are removed from the gas, and are sold as fertilizers.