Instead, the most serious health consequences related to NOx exposure occur indirectly, when nitrogen dioxide reacts with other air pollutants to form photochemical smog and acid rain.
Methods for controlling the release of nitrogen oxides to the atmosphere fall into three major categories: precombustion, combustion, and postcombustion systems.
The goal of precombustion systems is to remove nitrogen from a fuel so that, once it is burned, it releases lower concentrations of nitrogen oxides to the atmosphere.
One method for achieving this objective is hydrodenitrogenation, a process in which hydrogen gas is mixed with a liquid fuel and then heated gently over a catalyst, such as nickel-molybdenum or nickel-vanadium.
These conditions encourage nitrogen compounds in the fuel to react with hydrogen to form ammonia, which can be removed and reused:
N2 + 3H2→ 2NH3
The fuel that remains contains less nitrogen and burns “more cleanly,” that is, with the release of fewer nitrogen oxides.
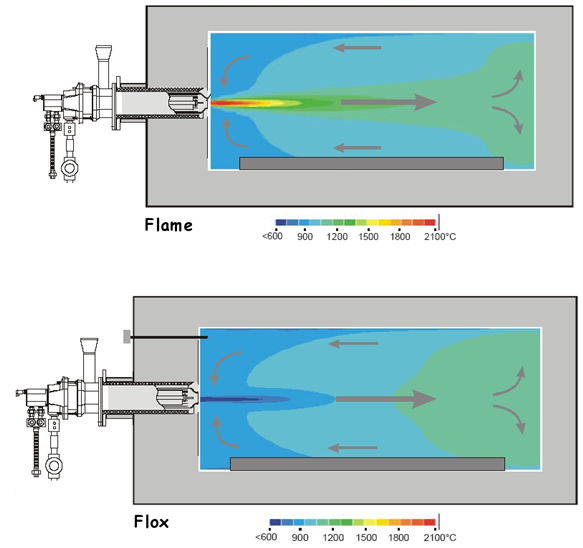
Combustion systems aim to reduce the release of nitrogen oxides by lowering the combustion temperature.
Because the rate at which nitrogen and oxygen react is very temperature sensitive, reducing the combustion temperature can also reduce the rate at which nitrogen oxides are produced.
The problem with this approach is that reducing combustion temperature also reduces the efficiency of fuel combustion. Therefore, more the effective the process for reducing nitrogen oxide emissions, lesser will be the efficiency of the combustion of fuel in a furnace or an automotive engine.
The challenge is to find some mechanism that achieves a reasonable compromise between these two goals, maintaining the efficiency of fuel combustion while reducing the emission of nitrogen oxides.
A number of systems have been devised to meet these two competing objectives.
In one approach, water is mixed with oil before it is sprayed into a combustion chamber. The presence of water reduces combustion temperatures enough to reduce nitrogen oxide emissions by up to 15 percent without significantly reducing combustion efficiency.
Another approach is to reduce combustion temperatures by using cool air in the combustion chamber, adding cool inert gases to the chamber, or recycling cool exhaust gases through the chamber.
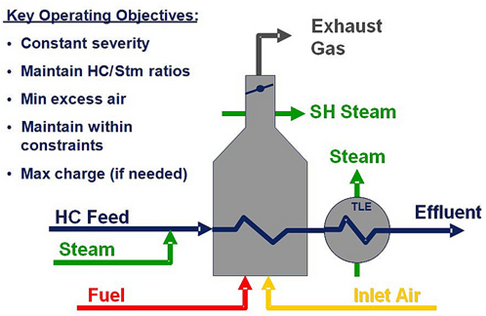
One of the most effective methods for controlling NOx production in combustion chambers is called low-excess-air firing. As the name suggests, this approach makes use of just enough air to allow combustion of the fuel, reducing to a bare minimum the amount of oxygen available for the conversion of nitrogen to NOx.