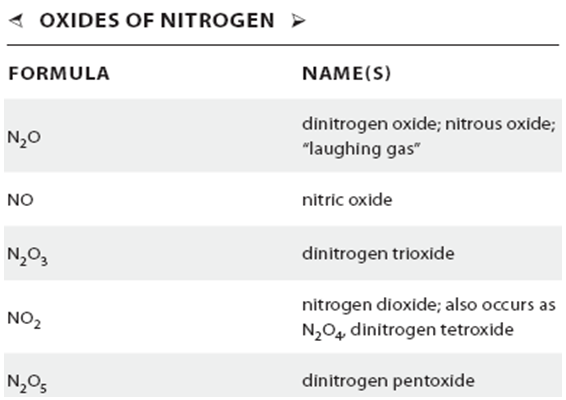
Oxides of Nitrogen
Nitrogen reacts with oxygen to form five oxides. The two most important with regard to air pollution are NO and NO2, which are sometimes referred to together as NOx.
Nitrogen and oxygen coexist in the atmosphere without reacting to any significant extent. At room temperature, for example, air normally consists of no more than about 0.000,000,000, 1 (1 × 10–10) ppm of
NO, formed in the following reaction:
N2 + O2→ 2NO
The reaction rate for this reaction accelerates rapidly, however, with increase in temperature.
For example, at about 1,000°C, the concentration of NO reaches about 100 ppm and at about 1,500°C, it reaches nearly 1,000 ppm.
The most common source of nitrogen oxides, therefore, is high temperature combustion processes.
Residential sources, such as gas stoves and home heaters, are also responsible for the release of significant amounts of NOx into the atmosphere.
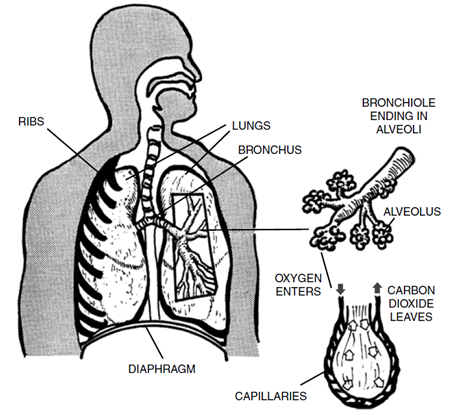
The primary health effect of nitric oxide results from its tendency to react with hemoglobin in red blood cells, in much the same way as carbon monoxide does.
The concentration of nitric oxide is normally so low, however, that this effect is relatively minor, especially compared with the effects of carbon monoxide exposure.
By contrast, nitrogen dioxide is a highly toxic gas with potentially serious effects on the human respiratory system, depending on the degree of exposure.
Exposures to low concentrations of the gas for short periods (less than three hours) typically produces relatively modest effects, including changes in airway responsiveness and lung function in individuals who have a history of respiratory disorder.
Children under the age of 12 are more likely to experience respiratory disorders under such conditions.
At higher concentrations, the effects are often more serious. Exposures to concentrations of 50–100 ppm for periods of six to eight weeks may cause inflammation of lung tissue.
Exposure to greater concentrations (150–200 ppm) for shorter periods may result in a disease known as bronchiolitis fibrosa obliterans, which can be fatal within three to five weeks.
The concentration of nitrogen oxides found in the air is almost always much less than these levels, and the health effects described here rarely occur except in accidents or spills in which nitrogen dioxide is released to the air.