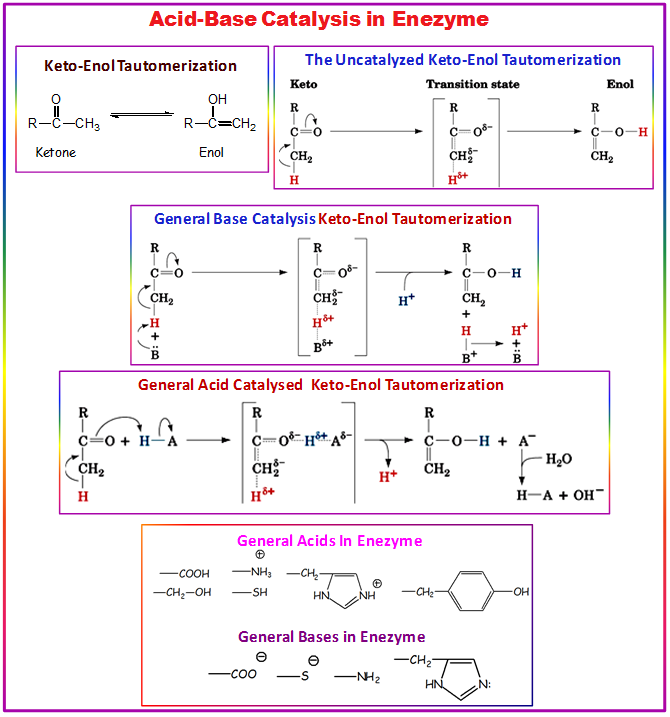
3.5.1. Acid-Base Catalysis: Catalysis Involving Proton Donors or Acceptors
- Proton donors and acceptors, i.e. acids and bases, may donate and accept protons in order to stabilize developing charges in the transition state.
- This has the effect of activating nucleophile and electrophile groups, or stabilizing leaving groups. Histidine is often the residue involved in these acid-base reactions, since it has a pKa close to neutral pH and can therefore both accept and donate protons.
- Many reaction mechanisms involving acid/base catalysis assume a substantially altered pKa. This alteration of pKa is possible through the local environment of the residue.
- The pKa is can be modified significantly by the environment, to the extent that residues which are basic in solution may act as proton donors, and vice versa.
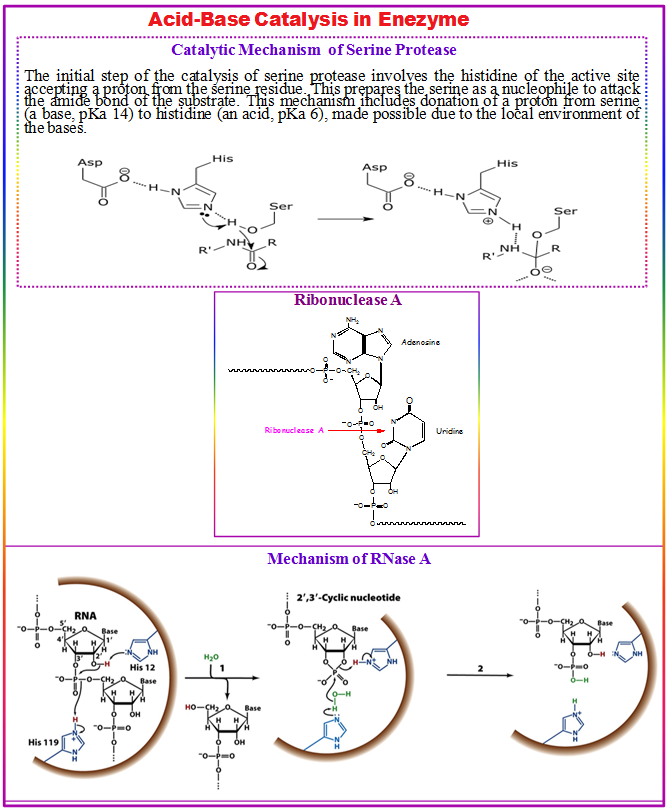
- The catalytic effect of the above example is mainly associated with the reduction of the pKa of the oxy anion and the increase in the pKa of the histidine, while the proton transfer from the serine to the histidine is not catalyzed significantly, since it is not the rate determining step.