3.2.3. How Enzymes work?
- Many reactions in biochemistry are spontaneous, i.e. they are thermodynamically favorable (ΔG < 0).
- The oxidation of glucose to produce carbon dioxide and water is thermodynamically favorable (ΔG°= -2870 kJ/mol). However, a jar of sugar in water is highly stable in the absence of microbial contamination.
- Biological reactions are almost always under kinetic control. Thus, a given amount of energy must be put into the system (energy of activation) in order for energy to be released from biochemical reactions. The rate of a reaction is related exponentially to the energy of activation.
- Two ways to effect the reaction are- i) raising the free energy of the substrates, or ii) decreasing the energy of activation for the reaction.
- Enzymes catalyze reactions by lowering the activation energy barrier.
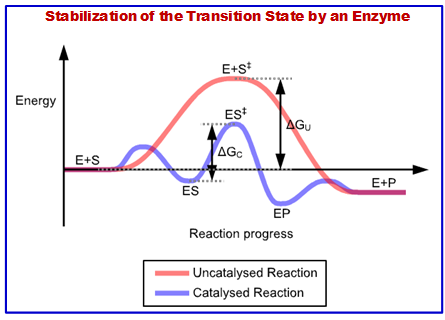
Figure 3.3: Energetics of stabilization of T.S. by an enzyme.
3.3. Advantages and Important Stereochemical Features of Enzyme Catalysis:
- Enzymes are natural proteins, available from renewable resources.
- Enzyme catalysis need: Mild Reaction Conditions – 37° C, near neutral pH and std. pressure.
- Enzymes are Biodegradable – waste management problems reduced.
- They are highly specific.
- Enzymes display three major types of selectivities:
- Chemoselectivity: Enzyme acts on a single type of functional group, other sensitive functionalities, which would normally react to a certain extent under chemical catalysis, survive. As a result, enzyme catalyzed reactions tend to be cleaner.
- Regioselectivity and diastereoselectivity: Enzyme, out of their complex three-dimensional structure, can distinguish between functional groups situated in different regions of the substrate molecule.
- Enantioselectivity: Enzymes are protein, made up of L-amino acids →, so, enzymes are chiral catalysts. So, any type of chirality present in the substrate molecule is recognized in the enzyme-substrate complex. Thus a prochiral substrate may be transformed into an optically active product and both enantiomers of a racemic substrate may react at different rates.
- Because of enantioselective nature, synthetic chemists have become interested in enzyme catalyzed enantioselective transformation.
- They are environmentally acceptable (‘GREEN’ ), as they are completely degraded in the environment.