3.2.2. Brief History of Enzymes
- In, 1833, the first enzyme was isolated from aqueous extract of malt added ethanol. The heat-labile precipitate that is now known as amylase was utilized to hydrolyze starch to soluble sugar.
- In 1878, Kühne coined the term enzyme which means “in yeast”.
- In 1898, Duclaux proposed that all enzymes should have suffix “ase”.
- Thus, Enzymes was defined as natural proteins capable of catalyzing chemical reactions.
- Enzymes have molecular weights of several thousand to several million, yet it can catalyze transformations on molecules as small as carbon dioxide and nitrogen.
- Enzymes function by lowering transition-state energies and energetic intermediates and by raising the ground-state energy.
- First enzyme recognized as protein was jack bean urease, crystallized in 1926 by James B. Sumner of Cornell University. He later, in 1946, received the Nobel Prize for his work with the enzyme urease.
- Urease is an enzyme that catalyzes the conversion of urea to ammonia and carbon dioxide. Certain bacteria that convert urea to ammonia as part of the nitrogen cycle contain this enzyme.
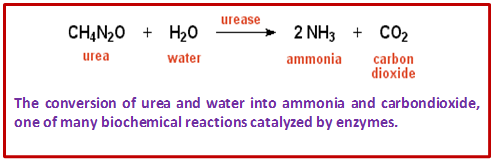
Figure 3.2: Ureas enzyme catalyzed conversion of urea to ammonia
- Almost all Enzymes are proteins. Their ability to catalyze reactions is attributable to their primary, secondary, tertiary, and quaternary structures.
- As catalysts, enzymes are both efficient and highly specific for a particular chemical reaction.
- Cofactors are involved in reactions along with enzyme where molecules are oxidized, reduced, rearranged or connected.
3.2.3. Characteristic of Enzyme Catalysis
- Almost all biochemical processes are catalyzed by enzymes.
- As almost all Enzymes are proteins, their ability to catalyze reactions is attributable to their primary, secondary, tertiary, and quaternary structures.
- Enzymes have a high degree of specificity for their substrates.
- Enzymes accelerate chemical reactions tremendously.
- Enzymes can function in aqueous solution under mild conditions, which are unlike the conditions that are frequently needed in organic chemistry.
- Enzymes are effective in minute amounts because they are not used up in the reaction that they catalyze.
- Enzymes do not affect the direction of the reaction but make the reaction reach equilibrium sooner.
- Both synthesis and decomposition of molecules in a living system normally proceed too slowly to be useful to metabolic survival. However, the presence and activity of enzymes speed up those to support life activities of cellular metabolism.
- Enzymes make up a substantial portion of the total protein of the cell. A typical cell contains about 3000 different kinds of enzyme molecules and many copies of each kind.
- Within a cell, chemical reactions take place within a narrow temperature and pH range. This is possible because enzymes generally lower the activation energy of a reaction through a variety of mechanisms.
- Enzymes necessarily are very large, because,
- Most effective binding of substrate results from close packing of atoms within enzymes.
- Remainder of enzyme outside active site is required to maintain integrity of the active site.
- May serve to channel the substrate into the active site.
- Active site aligns the orbitals of substrates and catalytic groups on the enzyme optimally for conversion to the transition-state structure-- called orbital steering.