3.1. Introduction to Enzyme Catalysis and Kinetics
3.1.1. Catalysis
Catalysis is the change in rate of a Chemical/Biochemical reaction due to the change in activation energy of that reaction by the involvement of a catalyst. A catalyst by nature and its definition, is not consumed by the reaction unlike other reagents participate in the chemical reaction. A catalyst may participate in multiple chemical transformations. Acceleration of the reaction rate is called positive catalysis and the catalysts speed up the same is called positive catalyst. On the other hand, substances that interact with catalysts to slow the reaction are called inhibitors or negative catalysts and the process is known as negative catalysis. Substances that increase the activity of catalysts are called promoters, and substances that deactivate catalysts activity are called catalytic poisons.
A catalyst generally brings down the energy of the activated state and thus the catalytic reactions have a lower rate-limiting free energy of activation than the corresponding unanalyzed reaction, resulting in higher reaction rate at the same temperature. However, the mechanistic explanation of catalysis is complex. Catalysts affect the reaction environment favorably by binding to the reagents to polarize bonds.
3.1.2. Catalysis and Reaction Energetic
In general, catalysts work by providing an alternative pathway which involves a different transition state of lower activation energy compared to an unanalyzed reaction. Consequently, more molecular collisions have the energy needed to reach the transition state. Hence, catalysts enable reactions that would otherwise be blocked or slowed by a kinetic barrier. The catalyst can increase reaction rate. It can enhance selectivity, or the reaction proceeds at lower temperatures in presence of a catalyst.
In the catalyzed elementary reaction, catalysts do not change the extent of a reaction. Thus, they have no effect on the chemical equilibrium of a reaction because the rate of both the forward and the reverse reaction are affected equally. This fact is a consequence of the second law of thermodynamics. If a catalyst changes the equilibrium, then it must be consumed as the reaction proceeds, and thus it is also a reactant. The catalyst stabilizes the transition state more than it stabilizes the starting material. Therefore, it decreases the kinetic barrier by decreasing the difference in energy between starting material and transition state (Figure 3.1).
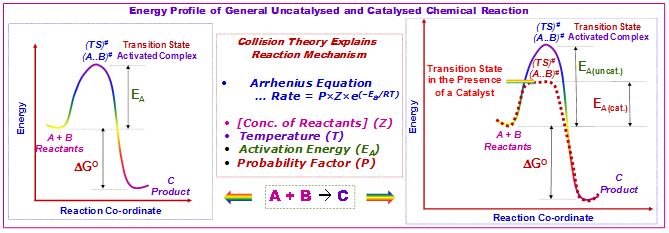
Figure 3.1: Potential energy diagram showing the effect of a catalyst in a hypothetical exothermic chemical reaction A + B to give product C. The presence of the catalyst opens a different reaction pathway with lower activation energy.
3.1.3. Types of Catalysis
Catalysts can be either heterogeneous or homogeneous, depending on whether a catalyst exists in the same phase as the substrate. Following are the main types of catalysts.
- Heterogeneous catalysts
- Homogeneous catalysts
- Electro catalysts
- Organocatalysis
- Biocatalysts (enzymes)
3.2. Biocatalysis: The Enzyme as a Catalyst
3.2.1. Definition
Biocatalysis is the use of natural catalysts, such as a catalytic protein which is most of the time referred to as an enzyme, to perform chemical transformations on organic compounds or biochemical reaction inside the living cells. Catalysis of biochemical reactions in the cell is vital due to the very low reaction rates of the unanalyzed reactions.