Mechanism of single ion exchange : Single ion exchange takes place when in the solid-solution system only two ions interact. If ion A+ on the solid Z is to be replaced by ion B+ present in the solution then in simple case of two monovalent cations, the exchange equilibrium can be written as
![]()
The subscripts ‘s’ and ‘z’ represent solution and solid respectively. In case of ideal exchange, that is both the exchanger and solutions are ideal with single type of adsorption sites, the equilibrium constant Ka can be written as
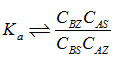

Therefore it follows that
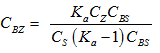
If it is not an ideal system the above equation must be expressed in terms of concentration and activity coefficients.
Example: Na+ ions in NaY Zeolites can be replaced by NH4+ ions. NaY zeolite [Na2O.Al2O3. 5 SiO2] contains 9.9 wt% Na and 73% Na can be exchanged.
Book References :
• K.P. de Jong. , Synthesis of solid catalysts , Wiley –VCH, 2009
• J.T. Richardson, Principle of catalysts development, Plenum Press, 1989
• G. Ertl, H. Knozinger & J. Weitkamp, Handbook of Heterogeneous Catalysis Vol 1, Wiley – VCH, 1997
• R. J. Farrauto & C. H. Bartholomew, Fundamentals of Industrial Catalytic Processes, Blackie Academic & Professional, 1997