In case of dry impregnation, in addition to diffusion and adsorption processes, another phenomenon occurs, which is the pressure driven capillary flow of the solution inside the empty pores. This can be represented by Darcy's law. An important parameter from introduction of Darcy law is the solution viscosity ‘μ'. In case of aqueous solution and in the common range of concentration used for impregnation, viscosity increases almost proportionally with concentration. It also increases with the presence of organic ligands attached to the metal ions. Viscosity and concentration have opposite effects on precursor diffusion; a high concentration tends to favor the diffusion of the solute towards the centre of the pellet, while a high viscosity tends to hinder the diffusion.
Drying
Impregnation is followed by elimination of the solvent. The impregnated sample is heated in an oven in flow of gas as discussed earlier. The gas may be air, oxygen, nitrogen or any other gas depending on the requirement. The temperature is generally maintained slightly higher than the boiling point of the solvent e.g 110-120°C for water. The elimination of water from the pores leads to the increase of precursor concentration up to saturation and consequent crystallization, preferably on the seeds resulting from the interaction with the support. Apart from temperature, heating rate affects the drying process.
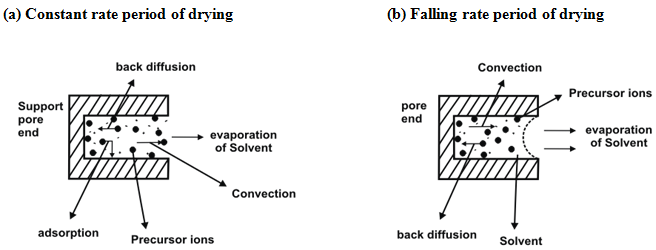
Fig. 2. Schematic representation of drying process during impregnation method