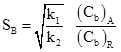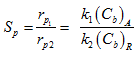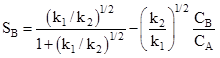If differences in diffusivities are neglected ![]()
|
(8) |
In absence of internal mass transfer resistance selectivity of B is given as
|
(9) |
Comparison of equations (8) and (9) shows that the intrapellet diffusion resistance reduces the selectivity.
(2) For parallel irreversible first order reaction with same reactant. B is desired.
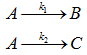
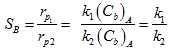 .
.
Hence at this conditions selectivity of B is unaffected by internal transport effect.
(3) For irreversible first order consecutive reaction
![]()
Here B is the desired product. The selectivity of product B with respect to reactant A is can be expressed as
|
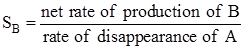 |
or |
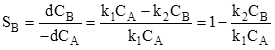 |
(10) |
The CA and CB are bulk concentrations of A and B respectively. The equation (10) gives selectivity of B for whole pellet provided that the internal diffusion resistance is negligible. In presence of significant intrapellet diffusion resistance, the concentration of CA and CB changes within the pellet and the selectivity will vary with the position within the pellet as CB/CA changes. Diffusion resistance causes CA to decrease from outer surface to the center of pellet. The product B is formed within pellet and has to diffuse out from the pellet into bulk. Hence concentration of B increases toward center of pellet due to resistance. Hence from equation (10) it can be seen that decrease of CA and increase of CB toward center of pellet result in reduction of pellet selectivity for B from centre to outer surface. For the given first order reaction in presence of strong diffusion resistance (say ![]() ) and assuming equal effective diffusivities, the selectivity of B can be obtained as
) and assuming equal effective diffusivities, the selectivity of B can be obtained as
|
(11) |
Comparison of equations (10) and (11) shows that the selectivity is significantly reduces when internal diffusional resistance is significant.
Book Reference :
• J. M. Smith, Chemical Engineering Kinetics , McGrawHill Book Company, 1981
• H. S. Fogler, Elements of Chemical reaction engineering , Prentice Hall of India, 1999
• J.J. Carberry , Chemical and catalytic reaction Engineering, Dover Publications, 2001
• O. Levenspiel , Chemical reaction engineering, John Wiley & sons, 1995