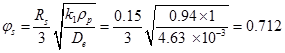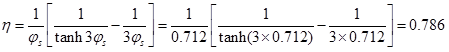Solved examples:
Dehydrogenation of hydrocarbon (mol.wt = 58) at atmospheric pressure was carried out over chromia-alumina catalysts at 5300C. The reaction follows a first order kinetics with rate constant of 0.94 cm3/s. g cat. The spherical catalysts are of 30 mm diameter and average pore radius is 11 nm. Pellets have porosity of 0.35 and density of 1 gm/cm3. Assume Knudsen diffusivity to be dominant. Predict an effectiveness factor for the catalysts. Use parallel pore model with a tortuosity factor of 3.0
Knudsen diffusivity is given as : 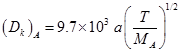
Solutions:
Density of catalysts pellets = 1 gm/cm3, T= 5300C; Dp = 0.3 cm ; Rs = 0.3/2 = 0.15 cm
k1 = 0.94 cm3/s. g cat ; a=pore radius = 11 nm = 11 × 10-7 cm
δ = 3 ; ε = 0.35
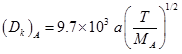
MA =58 ; a = 11 nm = 11 × 10-7 cm ; T= 5300C = 530 + 273 = 803 K
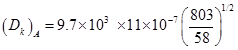 = 0.0397 cm2/s
= 0.0397 cm2/s
![]()
D=DK= 0.0397 cm2/s ; δ = 3, ε = 0.35
![]() = 4.63 ×10-3 cm2/s
= 4.63 ×10-3 cm2/s