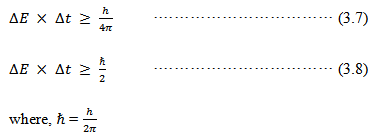Absorption peaks and line widths: Absorption of radiation is the first step in any spectroscopic experiment. Absorption spectra are routinely recorded for the electronic, rotational, and vibrational spectroscopy. It is therefore important to see |
 |
how an absorption spectrum looks like. As we have already seen, a transition between states takes place if the energy provided by the electromagnetic radiation equals the energy gap between the two states i.e.
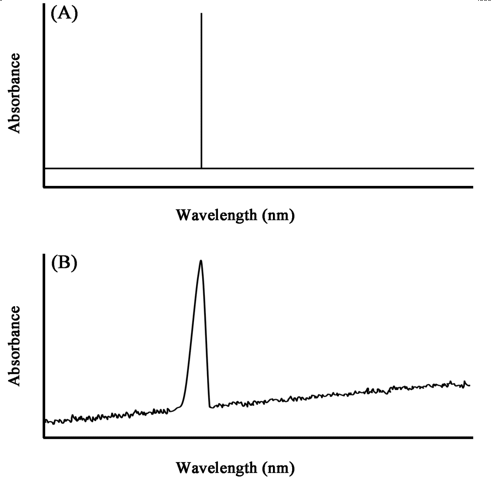
Figure 3.6 An idealized spectrum for a single wavelength transition (A) and an experimentally obtained spectrum (B) In practice, however, the absorption lines are not sharp but appear as fairly broad peaks (Figure 3.6B) for the following reasons: |
|
- Instrumental factors: The slits that allow the incident light to impinge on the sample and the emerging light to the detector have finite widths. Consider that the transition occurs at wavelength, λt. When the wavelength is changed to λt+ ∆λ or λt– ∆λ , the finite slit width allows the radiation of wavelength, λt to pass through the slits and a finite absorbance is observed at these wavelengths. The absorption peaks are therefore symmetrical to the line at λ = λt.
- Sample factors: Molecules in a liquid or gaseous sample are in motion and keep colliding with each other. Collisions influence the vibrational and rotational motions of the molecules thereby causing broadening. Two atoms/molecules coming in close proximity will perturb the electronic energies, at least those of the outermost electrons resulting in broadening of electronic spectra. Motion of molecules undergoing transition also causes shift in absorption frequencies, known as Doppler broadening.
- Intrinsic broadening: Intrinsic or natural broadening arises from the Heisenberg's uncertainty principle which states that the shorter the lifetime of a state, the more uncertain is its energy. Molecular transitions have finite lifetimes, therefore their energy is not exact. If ∆t is the lifetime of a molecule in an excited state, the uncertainty in the energy of the states is given by: