Quantization of energy:
As put forward by Max Planck while studying the problem of Blackbody radiation in early 1900s, atoms and molecules can absorb or emit the energy in discrete packets, called quanta (singular: quantum). The quantum for electromagnetic energy is called a photon which has the energy given by equation 3.6. A molecule can possess energies in different forms such as vibrational energy, rotational energy, electronic energy, etc. Introduction to the structure of an atom in a General Chemistry course mentions about the electrons residing in different orbits/orbitals surrounding the nucleus, typically the first exposure to the discrete electronic energy levels of atoms. In much the same way, rotational and vibrational energy levels of molecules are also discrete. A molecule can jump from one energy level to another by absorbing or emitting a photon of energy that separate the two energy levels (Figure 3.3).
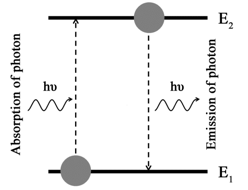
Figure 3.3 Transitions of a molecule between energy levels, E1 and E2 by absorbing/emitting the electromagnetic radiation