Energy of electromagnetic radiation:
Energy of an electromagnetic radiation is given by
![]()
where h is Planck's constant and has a value of 6.626 × 10-34 m2·kg·s-1. Based on the energy, electromagnetic radiation has been divided into different regions. The region of electromagnetic spectrum human beings can see, for example, is called visible region or visible spectrum. The visible region constitutes a very small portion of the electromagnetic spectrum and corresponds to the wavelengths of ~400 – 780 nm (Figure 3.2). The energy of the visible spectrum therefore ranges from ~2.5 × 10-19 to ~5 × 10-19 Joules. It is not convenient to write such
small values of energy; the energies are therefore written in terms of electronvolts (eV). One electronvolt equals 1.602 × 10-19 Joules. Therefore, the energy range of the visible spectrum is ~1.6 – 3.1 eV. Spectroscopists,
however, prefer to use wavelength (λ) or frequency (ν) or wavenumber![]() instead of energy.
instead of energy.
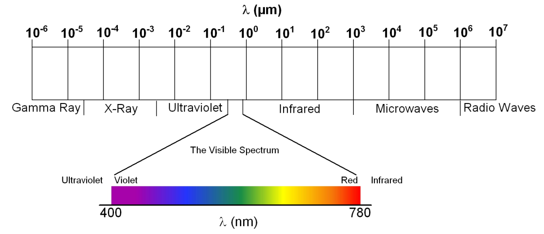
Figure 3.2 Electromagnetic spectrum