Electromagnetic spectrum and the atomic/molecular processes:
Molecules undergo processes like rotation, vibration, electronic transitions, and nuclear transitions. The energies underlying these processes correspond to different regions in the electromagnetic spectrum (Figure 3.4):
- Radiofrequency waves: Radiofrequency region has very low energies that correspond to the energy differences in the nuclear and electron spin states. These frequencies, therefore, find applications in nuclear magnetic resonance and electron paramagnetic resonance spectroscopy.
- Microwaves: Microwaves have energies between those of radiofrequency waves and infrared waves and find applications in rotational spectroscopy and electron paramagnetic resonance spectroscopy.
- Infrared radiation: The energies associated with molecular vibrations fall in the infrared region of electromagnetic spectrum. Infrared spectroscopy is therefore also known as vibrational spectroscopy and is a very useful technique for functional group identification in organic compounds .
- UV/Visible region: UV and visible regions are involved in the electronic transitions in the molecules. The spectroscopic methods using UV or visible light therefore come under ‘Electronic spectroscopy'.
- X-ray radiation: X-rays are high energy electromagnetic radiation and causes transitions in the internal electrons of the molecules.
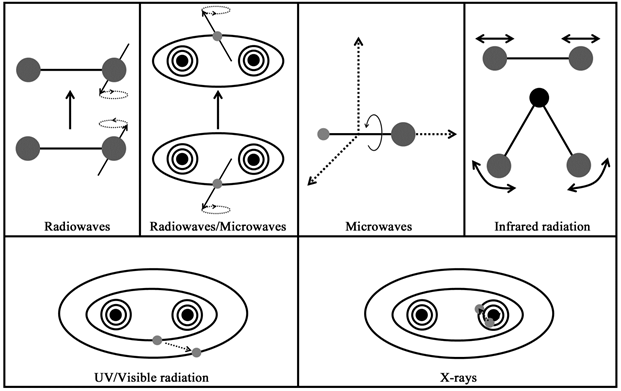 Figure 3.4 The range of atomic/molecular processes the electromagnetic radiation is involved in.
Figure 3.4 The range of atomic/molecular processes the electromagnetic radiation is involved in.