Mechanisms of interaction of electromagnetic radiation with matter:
In order to interact with the electromagnetic radiation, the molecules must have some electric or magnetic effect that could be influenced by the electric or magnetic components of the radiation.
- In NMR spectroscopy, for example, the nuclear spins have magnetic dipoles aligned with or against a huge magnetic field. Interaction with radiofreqency of appropriate energy results in the change in these dipoles.
- Rotations of a molecule having a net electric dipole moment, such as water will cause changes in the directions of the dipole and therefore in the electrical properties (Figure 3.5A and B). Figure 3.5B shows the changes in the y-component of the dipole moment due to rotation of water molecule.
- Vibrations of molecules can result in changes in electric dipoles that could interact with the electrical component of the electromagnetic radiation (Figure 3.5C).
- Electronic transitions take place from one orbital to another. Owing to the differences in the geometry, size, and the spatial organization of the different orbitals, an electronic transition causes change in the dipole moment of the molecule (Figure 3.5D).
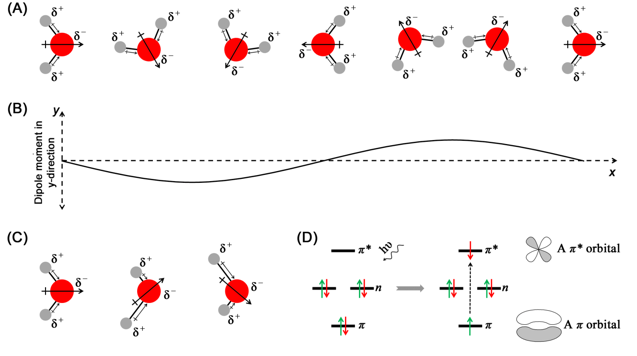
Figure 3.5 Panel A shows the rotation of a water molecule around its centre of mass. The change in the dipole moment as a result of rotation in plotted in panel B. Panel C shows the change in dipole moment of water due to asymmetric stretching vibrations of O—H bond. Panel D shows an electronic transition from π to π* orbital and the geometry of the two orbitals.
The above examples suggest that a change in either electric or magnetic dipole moment in a molecule is required for the absorption or emission of the electromagnetic radiation.