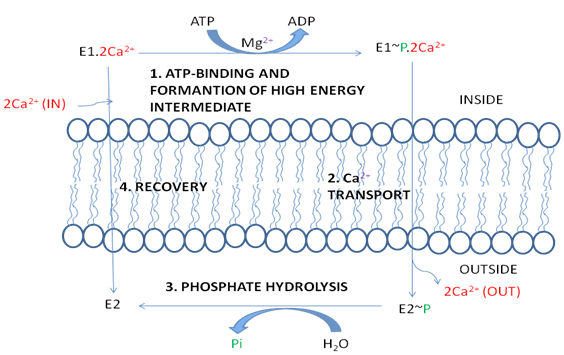
Figure 5: Scheme for the active transport of Ca2+ by the Ca2+ ATPase. Here (in) refers to the cytosol and (out) refers to the outside of the cell for plasma membrane Ca2+ ATPase or the lumen of the endoplasmic reticulum (or sarcoplasmic reticulum) for the Ca2+ ATPase of that membrane.
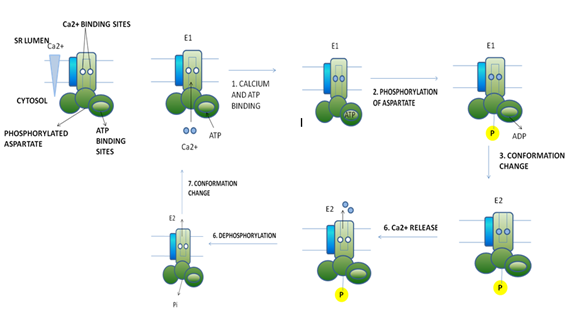
Figure 6: Operational model of the Ca2+ ATPase in the SR membrane of skeletal muscle cells. Only one of the two catalytic α subunits of this P-class pump is depicted. E1 and E2 are alternative conformations of the protein in which the Ca2+ binding sites are accessible to the cytosolic and exoplasmic faces, respectively. An ordered sequence of steps (1 – 6), as diagrammed here, is essential for coupling ATP hydrolysis and the transport of Ca2+ ions across the membrane. In the figure, ~P indicates a high-energy acyl phosphate bond; –P indicates a low-energy phosphoester bond.
Interesting facts:
- The X-ray crystal structure of lactose permease was first solved in 2003 by J. Abramson et al.
- Ouabain is a cardiac glycoside toxin. Potent inhibitors that bind to potassium binding sites. In the presence of Ouabain, Na+/K+ ATPase cannot return to its resting state.
- One major type of gradient linked active permeases is the sodium-glucose symport carrier.