Na+/K+ ATPase :
In mammalian cells, the Na+ and K+ gradients are the two major components of the electrochemical gradient across the plasma membrane. The cells maintain a lower intracellular Na+ concentration and higher intracellular K+ concentration with relative to extracellular space. Hence, for the generation and maintenance of the electrochemical gradients for Na+ and K+, it requires Na+/K+ ATPase, which is an ion pump that couples ATP hydrolysis to cation transport. It also helps to set the negative resting membrane potential, which regulates the osmotic pressure to avoid cell lysis. The Na+/K+ ATPase belong to P-class ATPase which is commonly found in the plasma membranes of higher eukaryotes. This transmembrane protein consists of two types of subunits: a 110-kD non-glycosylated α- subunit that contains the enzyme’s catalytic activity and binding sites for ATP, Na+ and K+ ions, and a 55-kD glycoprotein β-subunit of unknown function. The smaller β-subunit has one transmembrane domain that stabilizes the α-subunit and is important in membrane insertion. The α- subunit has eight transmembrane α-helical segments and two large cytoplasmic domains and the β- subunit has a single transmembrane helix and a large extracellular domain. The protein may function as an (αβ)2 tetramer in vivo.
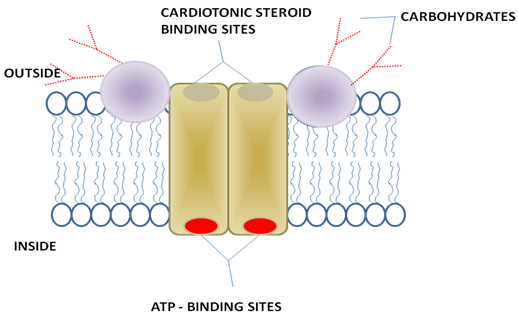
Figur 2: Na+/K+ ATPase. The diagram shows the transporter’s putative dimeric structure and its orientation in the plasma membrane. Cardiotonic steroids bind to the external surface of the transporter, thereby inhibiting transport.
The Na+/K+ ATPase is also called as the Na+/K+ pump because it pumps 3 Na+ out of and 2 K+ in both direction across the membrane in presence of hydrolysis of ATP. The overall reaction is:
3 Na+(in) + 2 K+(out) + ATP + H2O →3 Na+(out) + 2 K+(in) + ADP + Pi
The important feature to the Na+/K+ ATPase is the phosphorylation of a specific Asp residue of the transport protein which phosphorylates only in the presence of Na+, whereas the resulting aspartyl phosphate residue is subject to hydrolysis only in the presence of K+. Hence it has two conformations named E1 and E2. The protein appears to operate in the following (explained in figure 4):
- The protein in the E1 state has three high-affinities Na+ binding sites and two low-affinity K+ binding sites accessible to the cytosolic surface of the protein. Hence E1 binds three Na+ ions inside the cell and then binds ATP to yield an E1 .ATP.3 Na+ complex.
- ATP hydrolysis produces ADP and a “high-energy” aspartyl phosphate intermediate E1-P.3 Na+.
- This “high-energy” intermediate relaxes to its “low-energy” conformation, E1~P.3 Na+, and releases its bound Na+ outside the cell.
- E2-P binds two K+ ions from outside the cell to form an E2-P.2 K+ complex.
- The phosphate group is hydrolyzed, yielding E2 .2 K+.
- E2 .2 K+ changes conformation, releases its two K+ ions inside the cell, and replaces them with three Na+ ions, thereby completing the transport cycle.