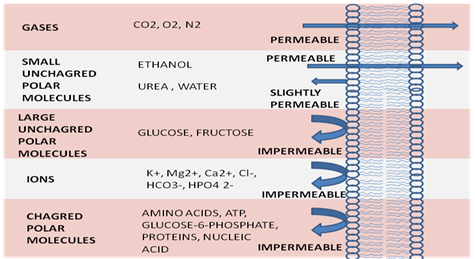
Figure 1: Relative permeability of a pure phospholipid bilayer to various molecules. A bilayer is permeable to small hydrophobic molecules and small uncharged polar molecules, slightly permeable to water and urea, and essentially impermeable to ions and to large polar molecules.
Thermodynamics of transport :
The diffusion of a substance A, across the two sides of a membrane thermodynamically resembles a chemical equilibration.
![]()
In the following sections, the free energy of a solute A, varies with its concentration:
![]()
where
GA is the chemical potential (partial molar free energy) of A (the bar indicates quantity per mole)
G°`A is the chemical potential of its standard state.
Thus, a difference arises in the concentrations of the substance on two sides of a membrane and generates a chemical potential difference:
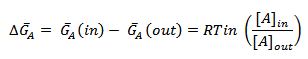
If the concentration of A outside the membrane is greater than that inside, ΔGA for the transfer of A from outside to inside will be negative and the spontaneous net flow of A will be inward. Conversely, if [A] is greater inside than outside, ΔGA is positive and an inward net flow of A can occur only if an exergonic process, such as ATP hydrolysis, is coupled to it to make the overall free energy change.
The transmembrane movement of ions also depends in charge differences across the membrane, thereby generating an electrical potential difference which is given by:
![]()
where ΔA is termed the membrane potential. Consequently, if A is ionic, must be amended to include the electrical work required to transfer a mole of A across the membrane from outside to inside:
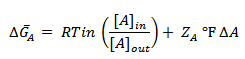
where
ZA is the ionic charge of A
F, the Faraday constant, is the charge of a mole of electrons (96,485 C /mol; C is the symbol for coulomb)
GA is now termed the electrochemical potential of A.
The membrane potentials of living cells are commonly as high as 100 mV (note that 1 V = 1 J /C).