The equality sign in Eq. (2.3.22) holds good for the energy equation based on first law of thermodynamics. However, the more appropriate form of second law of thermodynamics accounts for losses by means an inequality. It states that, the time rate of increase of the entropy of a system must be greater than or at least equal to the sum of ratio of net heat transfer rate into the system to absolute temperature for each particle mass in the system receiving heat from surroundings. This general statement can be written mathematically as,
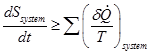 |
(2.3.23) |
At the instant, when the system and control volume are identical, the RHS of Eq. (2.3.23) may be written as,
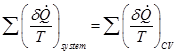 |
(2.3.24) |
Now, RTT can be applied to the variable entropy S and the corresponding intensive property becomes ![]() . For a fixed non-deforming control volume, the expression of RTT becomes,
. For a fixed non-deforming control volume, the expression of RTT becomes,
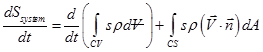 |
(2.3.25) |
Combination of Eqs (2.3.23, 2.3.24 & 2.3.25) gives,
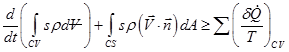 |
(2.3.26) |
Eq. (2.3.26) can be simplified for steady, one-dimensional flow with single inlet as,
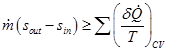 |
(2.3.27) |
Considering the specific entropy (s) and with infinitesimal small control volume at uniform absolute temperature (T), Eq. (2.3.27) is simplified as
(2.3.28) |
The equality sign holds good for any reversible (frictionless) process while the inequality sign is applicable for irreversible processes involving friction.