Energy Equation
The first law of thermodynamics for a system states that the rate of increase of the total stored energy of the system is equal to net rate of energy additions by the heat transfer into the system plus net rate of energy addition by work transfer into the system. The mathematical statement for energy equation is given by,
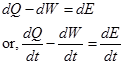 |
(2.3.12) |
Now, RTT can be applied to the variable energy E and the corresponding intensive property becomes ![]() . So, for a fixed control volume, energy equation is written as,
. So, for a fixed control volume, energy equation is written as,
(2.3.13) |
Here, Q is the energy transfer by heat and W is the energy transfer by work. They are considered as positive when heat is added to the system or work is done by the system. The system energy (per unit mass) mainly consists of different forms such as internal energy, kinetic energy and potential energy.
(2.3.14) |
The energy transfer by heat ![]() involves the mode of transfer i.e. conduction/convection/radiation. The time derivatives of work transfer can be represented as,
involves the mode of transfer i.e. conduction/convection/radiation. The time derivatives of work transfer can be represented as,
(2.3.15) |
The shear work due to viscous stresses ![]() and work done due to pressure forces
and work done due to pressure forces ![]() occur at the control surface while the shaft work
occur at the control surface while the shaft work ![]() is deliberately obtained by the system. Using Eq. (2.3.15) in (2.3.13), one can obtain the control volume energy equation.
is deliberately obtained by the system. Using Eq. (2.3.15) in (2.3.13), one can obtain the control volume energy equation.
(2.3.16) |
Here, the pressure work term is combined with the energy flux term because both involve surface integral. Introducing the thermodynamic property enthalpy ![]() that occurs in the fixed control volume, Eq. (2.3.16) becomes,
that occurs in the fixed control volume, Eq. (2.3.16) becomes,
(2.3.17) |