| |
Seebeck effect is a manifestation of the fact that if two points in a conductor (or a semiconductor) are maintained at different temperatures, the charged carriers (electorns or holes) in the hotter region, being more energetic (and, therefore, having higher velocities) will diffuse towards region of lower temperature. The diffusion stops when the electric field generated because of movement of charges has established a strong enough field to stop further movement of charges. For a metal, carriers being negatively charged electrons, the colder end would become negative so that Seebeck cofficient is negative. For a p-type semiconductor on the other hand, holes diffuse towards the lower temperature resulting in a positive Seebeck coefficient. |
| |
Performance of a thermocouple is determined by the Seebeck coefficient of the pair of metals forming the thermocouple. As it is impracticable to list the coefficient of all possible pairs, the Seebeck coefficients of metals are usually given with respect to Platinum as standard whose Seebeck coefficient is taken as zero. The following table gives the Seebeck coefficient (in 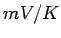 ) of some standard thermocouple material at 0 ) of some standard thermocouple material at 0 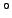 C. C. |
| |
| Material |
Seebeck |
Material |
Seeback |
Material |
Seebeck |
|
coefficient |
|
coefficient |
|
coefficient |
| Bismuth |
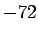 |
Lead |
4 |
Iron |
19 |
Constantan 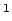 |
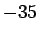 |
Tantalum |
4.5 |
Nichrome 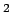 |
25 |
| Nickel |
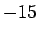 |
Rhodium |
6 |
Antimony |
47 |
| Potassium |
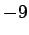 |
Gold |
6.5 |
Germanium |
300 |
| Sodium |
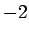 |
Silver |
6.5 |
Silicon |
440 |
| Mercury |
0.6 |
Copper |
6.5 |
Tellurium |
500 |
| Carbon |
3 |
Cadmium |
7.5 |
Selenium |
900 |
| Aluminium |
3.5 |
Tungsten |
7.5 |
|
|
|
| |
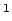 An alloy of copper (60%) and nickel (40%)
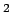 An alloy of Ni-Cr-Fe-Si |