Problems |
1) |
Calculate the bond order in hybridized CO and unhybridized CO. Extend the same analogy to hybridized and unhybridized NO, NO+ and NO - using the same energy level diagrams as those for CO. |
| |
|
| 2) |
What is the difference between a hybrid orbital and a molecular orbital ? Let  s be an s orbital with energy Es (ie Es = s be an s orbital with energy Es (ie Es =
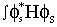 d d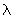 where H is the Hamiltonian operator for the atom) and Ep be the energy of where H is the Hamiltonian operator for the atom) and Ep be the energy of
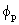 , a p orbital. If the hybrid orbital is Cs , a p orbital. If the hybrid orbital is Cs + Cp + Cp p, show that the energy of the hybrid orbital is Cs2 Es + Cp2 Ep. Note that p, show that the energy of the hybrid orbital is Cs2 Es + Cp2 Ep. Note that
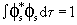 , ,
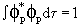 and and
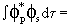 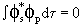 |
|
|
| 3) |
The dipole moment D is defined as the vector whose direction is from the -ve charge to the positive charge. |
|
|

|
| |
D = qr. q is the charge that is separated by distance r. For +e and -e separated by 1
 , D = 4.8 Debye = 4.8 x 10-18 esu.cm. The dipole moments of HF, HCl, HBr and CO are 1.9, 0.9, 0.7 and 1.05 Debye respectively and their bond lengths are 0.91, 1.27, 1.41 and 1.1 , D = 4.8 Debye = 4.8 x 10-18 esu.cm. The dipole moments of HF, HCl, HBr and CO are 1.9, 0.9, 0.7 and 1.05 Debye respectively and their bond lengths are 0.91, 1.27, 1.41 and 1.1
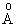 . Assuming that the excess or separated charges are localised at the nuclei, what are the charges that are separated in these molecules? . Assuming that the excess or separated charges are localised at the nuclei, what are the charges that are separated in these molecules? |
| |
|
| 4) |
Sketch the binding energy (vs internuclear distance r ) for CO. What is the reference (or zero) value of energy that you have to substract from the energy of CO to get the binding energy? |
| |
|
| 5) |
What are the electronic configuration of molecules HF, LiF and NO, CN and MgO? |
| |
|
| 6) |
KBr has a bond length of 2.94 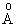 . If there is a separation of an unit of charge (i.e. electronic charge, e ) in KBr with the molecule behaving as K + Br-, what will be the dipole moment? Assuming that the repulsive forces between K + and Br- are very small compared to attractive forces, how much energy is needed to separate K+ from K + Br- at 2.94 . If there is a separation of an unit of charge (i.e. electronic charge, e ) in KBr with the molecule behaving as K + Br-, what will be the dipole moment? Assuming that the repulsive forces between K + and Br- are very small compared to attractive forces, how much energy is needed to separate K+ from K + Br- at 2.94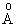 ? ? |
| |