We have studied in detail the previous lecture the shapes, sizes and contours of MOs of homonuclear diatomics. The main difference in heteronuclear diatomics is the "distortion" or the "tilting" of electron clouds towards the electronegative atoms. The 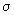 bond in LiF has been shown in fig 8.3(d) to be polarised towards the F atom. The bond in LiF has been shown in fig 8.3(d) to be polarised towards the F atom. The
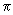 bonds formed by the sideways overlap of p- orbitals will also show the same distortion. The bonds in CO are sketched in fig 8.4. bonds formed by the sideways overlap of p- orbitals will also show the same distortion. The bonds in CO are sketched in fig 8.4.
|