39.6 |
Determination of the equilibrium constant of the reaction |
| |
If iodine is added to a moderately concentrated aqueous solution of KI, it combines with the iodide ion to form tri-iodide ion, 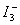 . Thus, in aqueous KI solution containing iodine, we have the chemical reaction . Thus, in aqueous KI solution containing iodine, we have the chemical reaction
KI + I2  KI3 and the equilibrium constant of the reaction is, KI3 and the equilibrium constant of the reaction is, |
| |
Since molecular iodine is soluble in both the aqueous and organic CCl4 phase, it obeys the distribution law whereas KI and KI3, being the electrolytes, are insoluble in CCl4. |
| |
Proceed as in previous exercise, but use aqueous KI solutions of different concentration in place of water. The aqueous layer will now have more I2, so the same thiosulphate (say 0.1N) may be used to titrate the two layers. The titre of aqueous layer correspond to total of free I2 and 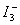 , and the concentration of free I2 may be calculated using the concentration in CCl4 layer and the previously determine K part. The initial I - concentration is known and since, , and the concentration of free I2 may be calculated using the concentration in CCl4 layer and the previously determine K part. The initial I - concentration is known and since, |
| |
Now, using values of (I2), (KI) and (KI3) in aqueous KI layer, the equilibrium constant can be calculated. |