| |
If in a system of two immiscible or slightly miscible solvent, a substance, soluble in both the solvents is added, then the added substance will distribute itself between the two liquids in a definite manner depending upon its solubility. At equilibrium the ratio of the concentrations of the solute in the two liquids is constant at a given temperature and is called the distribution coefficient or partition coefficient. It is important that the solute must exist in the two solvents in the same molecular state. If C, and C2 are the concentrations of the solute in the two solvents when equilibrium is reached, then at constant temperature. |
| |
Determine the distribution coefficient of iodine between carbon tetrachloride and water at a given temperature (or room temperature). |
| |
The molecular state of iodine in both the solvents CCl4 and water is the same as I2 and hence the partition coefficient is practically independent of concentration in dilute solutions. Hence, the distribution law in its simplest form may be applied i.e., 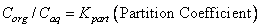 . . |
1. |
By means of a graduated pipette place about 50, 40, 30 and 20 mL of a saturated solution in I2 in CCl4 and properly labeled (1 to 4) glass stoppered bottles. Add approximately 0, 10, 20 and 30 mL CCl4 to bottle nos. 1, 2, 3 and 4, respectively. Add about 200 mL of distilled water to each of the bottles. |
2. |
Stopper the bottles tightly and shake thoroughly. Withdraw 20 mL of the aqueous layer carefully and titrate against 0.01 N Na2S2O3 using starch solution as indicator (repeat to concordance). To analyse the lower CCl4 layer, introduce a dry 5 mL pipette into the bottle while blowing continuously in and lower till the bottom in reached, withdraw 5 mL of the lower layer of CCl4 and titrate against 0.1N thiosulphate solution. Calculate the ratio of concentrations. |
| |
Similarly, titrate aqueous and CCl4 layer from bottle nos. 2, 3 and 4 and observe that the partition coefficient is reasonably constant. Take the mean value as K part.
While titrating the non-aqeuous layer, the titration flask must constantly be shaken, otherwise the equivalence point may pass without the disappearance of purple colour of non-aqueous layer. The addition of about 5mL of 10% KI solution also helps. KI facilitates extraction of iodine into aqueous solution during titration due to the formation of unstable complex ion 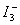 as as 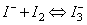 . .
|
| |
Distribution of succinic acid between water and ether could also be studied. Succinic acid also remains in normal molecular state in both water and ether. Take about 1.0, 1.5 and 2.0 g of succinic acid on three stoppered bottles numbered 1 to 3. To each bottle add about 50 mL ether and 50 mL distilled water. Titrate 10 mL of ethereal layer with 0.05 M NaOH and 10 mL g aqueous layer with 0.5 M NaOH.
Calculate the concentration of the acid in the two layers in terms of moles/ l and obtain the average value of he ratio of concentrations to get K part.
|
| |
An experiment using the distribution law is discussed below |