39.4 |
Excess Surface Energy (Surface Tension)
|
| |
In this experiment, we study the determination of surface concentration of 1-butanol in aqueous solution.
The addition of a surface active agent into any liquid changes the surface tension of a liquid. The change in surface tension is related to the excess surface concentration of the solution by the Gibb’s adsorption isotherm:
|
| |
where,
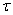 = surface excess concentration of solute per unit area = surface excess concentration of solute per unit area
R = gas constant
T = absolute temperature
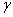 = surface tension of the solvent = surface tension of the solvent
c = concentration of the solute
|
| |
The surface tension may be compared with that of pure liquid using a Stalagmometer which is operated on the “drop-weight” principle. The Stalagmometer has a bulb with a capillary attached and marks on either side of the bulb. We let the fixed volume of a liquid flow through the capillary ad count the drops; knowing the densities of liquids, the drops weight and hence the surface tensions, may be compared: |
| |
where 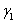 and and 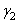 are the surface tension of water and liquid. d1 and d2 are the densities of water an liquid, respectively. n1 and n2 are the number of drops of water and liquid. are the surface tension of water and liquid. d1 and d2 are the densities of water an liquid, respectively. n1 and n2 are the number of drops of water and liquid.
|
| |
A 4% (v/v) solution of 1-butanol is supplied from which 3%, 2% and 1% solution of 1-butanol is prepared using dilution method. The Stalagmometer is clamped vertically. Water is sucked up into the Stalagmometer from a beaker to a level above the bulb. It is started to release and when the level crosses the mark, counting of the drops is started till the level crosses the lower mark. This experiment is repeated and the average of the reading is taken in calculation. The similar experiment is conducted with each diluted solution and their densities are determined using a specific gravity bottle. The value of
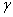 is plotted against ln c or the value of is plotted against ln c or the value of
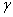 is plotted against c. From the plot, the slope at two point are found by drawing tangents at c = 1.5% and c = 2.5%. is plotted against c. From the plot, the slope at two point are found by drawing tangents at c = 1.5% and c = 2.5%.
The surface excess concentration of 1-butanol is calculated from the slope of the curve in each case. |