39.3 |
Experiment: to determine the average molecular weight of the polymer
|
| |
Viscosity of a polymer solution could be studied to determine average molecular weight of the polymer. An average molecular weight is calculated because the polymer molecules do not all have the same mass.
Empirically it has been found that the intrinsic viscosity is sensitive both to the shape and molecular weight of the macromolecular solute. The equation used by the Mark-Houwink relation is |
| |
where M is the average molecular weight and K and a are constant for a given solvent-solute system. The constant a is sensitive to the shape and varies from zero for hard sphere or 0.5 for random coils to 2.0 for rigid rods.
Variation of specific viscosity of a solution 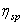 (specific viscosity or reduced viscosity) is defined as (specific viscosity or reduced viscosity) is defined as 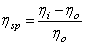 , where , where
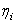 is the viscosity of solution i and is the viscosity of solution i and 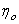 the viscosity of pure solvent) with the concentration of solute is given by the equation: the viscosity of pure solvent) with the concentration of solute is given by the equation:
|
| |
where, c is concentration of the polymer in g/mL and K is a constant.
The determination of average molecular weight of polyvinyl acetate in acetone and methanol uses the following Procedure:
Prepare solution of polyvinyl acetate in acetone containing approximately 1 mg/ mL. From this prepare four more solutions of 0.2, 0.4, 0.6, 0.8 mg/ mL by dilution. Measure the specific viscosity of all the four solutions as described above.
Repeat the above experiment with solution of polyvinyl acetate in methanol.
Plot 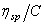 versus c and calculate versus c and calculate 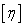 and K from equation (2). Also, using the empirical formula (1) and constant of K and a from the table given below, determine the molecular weight of polyvinyl acetate in these two solvents. and K from equation (2). Also, using the empirical formula (1) and constant of K and a from the table given below, determine the molecular weight of polyvinyl acetate in these two solvents.
|
| |
Solvent |
K |
a |
Acetone |
21.4 x 10 5 |
0.68 |
Methanol |
38 x 10 -5 |
0.59 |
|