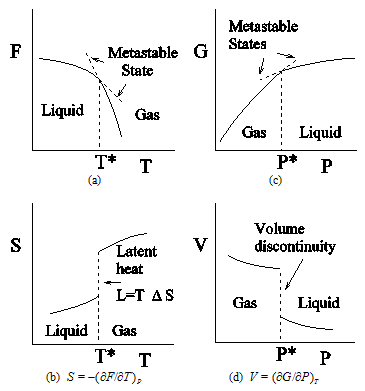
Figure 2.2: Variation of free energies and their first derivatives with respect to the respective parameters around a first order transition. (a) Plot of Helmholtz free energy F against temperature T at a constant pressure. (b) Plot of the entropy S, first derivative of F with respect to T, against T.
(c) Plot of Gibb's free energy G against pressure P at a constant temperature. (d) Plot of the volume V, first derivative of G with respect to P, against P. Discontinuities in entropy S (latent heat) as well as in volume V are marks of a first order transition.
First order transition often (not always) ends up at a critical point where second order transition takes place. In Fig.2.3, the characteristic behaviour of second order phase transitions is shown for a fluid system. At a first order phase transition the free energy curves of the two phases meet with a difference in slopes whereas at a second order transition the two free energy curves meet tangentially at the critical point ( ![]() ). The slopes of the curves changes continuously across the critical point. Therefore, there is no discontinuity either in entropy or in volume. Since there is no entropy discontinuity in second order transition, there is no emission (or absorption) of latent heat in this transition. It is a continuous phase transition where the system goes continuously from one phase to another without any supply of latent heat.
). The slopes of the curves changes continuously across the critical point. Therefore, there is no discontinuity either in entropy or in volume. Since there is no entropy discontinuity in second order transition, there is no emission (or absorption) of latent heat in this transition. It is a continuous phase transition where the system goes continuously from one phase to another without any supply of latent heat.
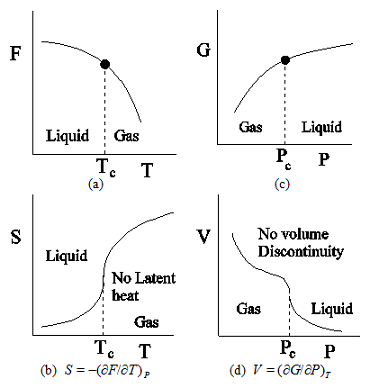
Figure 2.3: Variation of free energies and their first derivatives with respect to the respective parameters around a second order transition. (a) Plot of Helmholtz free energy F against temperature T at a constant pressure. (b) Plot of the entropy S, the first derivative of F with respect to T, against T. (c) Plot of Gibb's free energy G against pressure P at a constant temperature. (d) Plot of the volume V, the first derivative of G with respect to P, against P. No discontinuity is present in either entropy S or volume V.
Not only the first derivatives but also the second derivatives of the free energy show a drastic difference in their behaviour around the transition point in the first and second order transitions. For example, the specific heat ![]() diverges in the first order transition whereas in the second order transition specific heat has a finite discontinuity or logarithmic divergence at the critical point. Infinite specific heat in first order transition can be easily visualized by considering boiling of water. Any heat absorbed by the system will drive the transition (100°C water to 100°C steam) rather than increasing the temperature of the system. There is then an infinite capacity of absorption of heat by the system. In the second order phase transition, the response functions, the second derivatives of the free energy functions, are expected to diverge at the critical point. For example, the isothermal compressibility
diverges in the first order transition whereas in the second order transition specific heat has a finite discontinuity or logarithmic divergence at the critical point. Infinite specific heat in first order transition can be easily visualized by considering boiling of water. Any heat absorbed by the system will drive the transition (100°C water to 100°C steam) rather than increasing the temperature of the system. There is then an infinite capacity of absorption of heat by the system. In the second order phase transition, the response functions, the second derivatives of the free energy functions, are expected to diverge at the critical point. For example, the isothermal compressibility 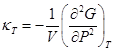 of a fluid system diverges at the critical point.
of a fluid system diverges at the critical point.