1. Introduction:
A phase is a state of matter in thermodynamic equilibrium. The same matter (or system) could be in several different states or phases depending upon the macroscopic condition (Temperature, Pressure, etc.) of the system. Different phases of water are our everyday experience. Ice, water and steam are the different states or phases of a collection of large number of H2O molecules. Given a macroscopic condition, the system spontaneously goes to a particular phase corresponding to lowest free energy as shown in Figure 2.1. For a closed system which exchange only energy with the surroundings has lowest Helmholtz free energy (F) at equilibrium and for an open system which exchange energy as well as mass with the surroundings the equilibrium corresponds to lowest Gibb's free energy (G). The free energy of a system is the sum of various energies associated with a collection of large number of atoms or molecules. Beside the kinetic energies of the particles, the potential energies due to inter atomic (or molecular) interactions contribute mostly to the free energy. Essentially, the different phases of matter are a consequence of interaction among a large number of atoms or molecules at a given thermodynamic condition.
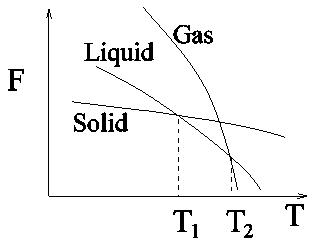
Figure 2.1: Variation of Helmholtz free energy F against temperature T at a constant pressure for a system in solid, liquid and gaseous phases. After T1, the system spontaneosly goes to the liquid phase.
There is a wide variety of phase transitions starting from liquid to gas, paramagnet to ferromagnet, normal to super conductor, liquid to liquid crystal and many other transitions. Most phase transitions belong to one of the two types - first order and second order phase transition. As per Ehrenfest's criteria, nth order phase transition corresponds to the discontinuity of the nth derivative of the free energy functions. Thus, in a first order transition, the first derivative of the free energy becomes discontinuous whereas in the second order phase transition, the second derivative of the free energy becomes discontinuous or diverges at the transition point. First and second order phase transitions then can be understood qualitatively in terms of discontinuity of free energy functions.