The response functions are not all independent. One could show for a fluid system,
![]()
where ![]() , the thermal expansion coefficient and similarly for a magnetic system,
, the thermal expansion coefficient and similarly for a magnetic system,
![]()
where ![]() . Since, the specific heat and compressibility are positive, it could be shown from the above relations that
. Since, the specific heat and compressibility are positive, it could be shown from the above relations that ![]() and
and ![]() . The equality holds either at
. The equality holds either at ![]() or at
or at ![]() , for example,
, for example, ![]() for water at
for water at ![]() .
.
From thermodynamic relations, it is already known that
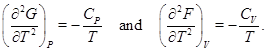
Since the specific heats are positive, these second derivatives are negative and as a consequence G and F both are concave functions of temperature T. It is also known that
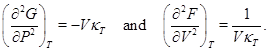
Since the compressibility is a positive quantity, G is a concave function of P whereas F is a convex function of V.
For magnetic systems, described by Hamiltonian ![]() , it can be shown that
, it can be shown that ![]() is a concave function of both T and H whereas
is a concave function of both T and H whereas ![]() is a concave function of T but a convex function of M.
is a concave function of T but a convex function of M.
3. First and second order transitions:
Both the Helmholtz free energy F and the Gibb's free energy G are concave function of temperature T and pressure P. However, F is a decreasing function of temperature T and G is an increasing function of pressure P. Variation of the Helmholtz free energy (F) with temperature T and the Gibb's free energy (G) with pressure P are shown in Fig.2.2 for a fluid system. Around a first order transition point ![]() , the free energy curves of the two phases meet with difference in slopes and both stable and metastable states exist for some region of temperature and pressure. At the transition temperature T*, the tangent to the curve F(T) versus T changes discontinuously and similarly for G(P) versus P , the tangent changes discontinuously at the transition pressure P*. The change in the slope of F with respect to T corresponds to entropy
, the free energy curves of the two phases meet with difference in slopes and both stable and metastable states exist for some region of temperature and pressure. At the transition temperature T*, the tangent to the curve F(T) versus T changes discontinuously and similarly for G(P) versus P , the tangent changes discontinuously at the transition pressure P*. The change in the slope of F with respect to T corresponds to entropy ![]() discontinuity. The change in the slope of G with respect to P corresponds to discontinuity in the volume
discontinuity. The change in the slope of G with respect to P corresponds to discontinuity in the volume ![]() . The transition is called first order because both entropy S and volume V are the first derivatives of the free energy functions and exhibit discontinuities. First order transitions are generally abrupt and are associated with an emission (or absorption) of the latent heat
. The transition is called first order because both entropy S and volume V are the first derivatives of the free energy functions and exhibit discontinuities. First order transitions are generally abrupt and are associated with an emission (or absorption) of the latent heat ![]() . Latent heat is released when the material cools through an infinitesimally small temperature change around the transition temperature. Most crystallization and solidification are first order transitions. For example, the latent heat
. Latent heat is released when the material cools through an infinitesimally small temperature change around the transition temperature. Most crystallization and solidification are first order transitions. For example, the latent heat ![]() Jg-1 comes out when water becomes ice. This happens sharply at 0° C temperature under the atmospheric pressure, when the H2 O molecules which wander around in the water phase gets packed in FCC ice structure releasing the excess energy as the latent heat. It can also be noted that in a first order phase transition there is generally a radical change in the structure of the material.
Jg-1 comes out when water becomes ice. This happens sharply at 0° C temperature under the atmospheric pressure, when the H2 O molecules which wander around in the water phase gets packed in FCC ice structure releasing the excess energy as the latent heat. It can also be noted that in a first order phase transition there is generally a radical change in the structure of the material.