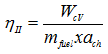When an S.S.S.F. chemical reaction takes place in such a way that both the reactants and products are in temperature equilibrium with the surroundings, the reversible work is given by
|
(3.117) |
where the ![]() s refer to the Gibbs function. The Gibbs function for formation,
s refer to the Gibbs function. The Gibbs function for formation,![]() is defined similar to the enthalpy of formation,
is defined similar to the enthalpy of formation,![]() The Gibbs function of each of the elements at 250 C and 1atm. is assumed to be zero, and the Gibbs function of each substance is found relative to this base. Table 3.4 gives
The Gibbs function of each of the elements at 250 C and 1atm. is assumed to be zero, and the Gibbs function of each substance is found relative to this base. Table 3.4 gives ![]() for some substance at 250 C, 1atm.
for some substance at 250 C, 1atm.
Second Law Efficiency of a Reactive System
For a fuel at T0, p0 the chemical exergy is the maximum theoretical work that could be obtained through reaction with environmental substances. However, due to various irreversibilities like friction and heat loss, the actual work obtained is only a fraction of this maximum theoretical work. The second law efficiency may thus be defined as the ratio of
|
(3.118) |
|
(3.119) |
The associated irreversibility and the consequent exergy loses require to be reduced to enhance the second law efficiency, which in turn, reduce the fuel consumption and also increases the cost. The trade off between the fuel savings and the additional costs must be carefully weighed.