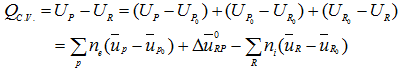First Law for Reactive Systems
For the S.S.S.F. process as shown in Fig. 3.4 the first law gives
HR + QC.V. = HP + WC.V. |
(3.97) |
or,
|
(3.98) |
or,
|
(3.99) |
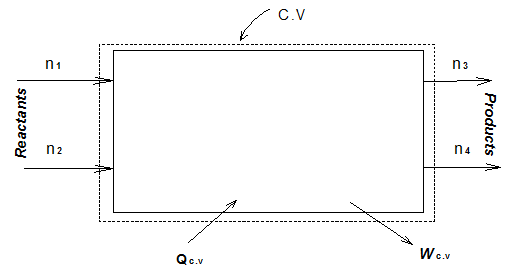
Fig.3.4 First law for a reaction system
When the states of reactants and products are not in the standard reference state (298K, 1atm), then, as shown in Fig. 3.5,
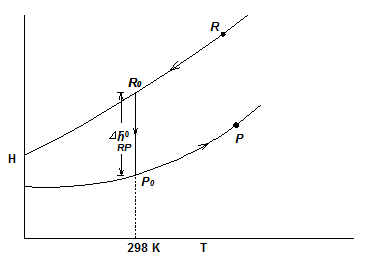
Fig. 3.5 Enthalpy of reactants and products varying with temperature
|
(3.100) |
|
(3.101) |
where ![]() is the enthalpy of reaction at the standard temperature (298 K). The variation of enthalpy with pressure is not significant.
is the enthalpy of reaction at the standard temperature (298 K). The variation of enthalpy with pressure is not significant.
|
(3.102) |
where ![]() is the internal energy of reaction at 298 K.
is the internal energy of reaction at 298 K.