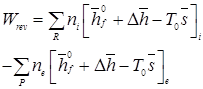Second Law Analysis of Reactive Systems
The reversible work for a steady state flow process, in the absence of changes in K.E. and P.E., is given by
Wrev = Σni(hi - T0si) - Σne(he - T0se) |
(3.113) |
For an S.S.S.F. process involving a chemical reaction
|
(3.114) |
The irreversibility for such a process is
|
(3.115) |
The availability, Ψ, in the absence of K.E. and P.E. changes, for an S.S.S.F. process is
Ψ = (h - T0s) - (h - T0s0) |
(3.116) |