The advection-diffusion equation becomes,
(38.23) |
The system will reach the equilibrium condition if sufficient time is available to the system. Then ∂S/∂t= 0
Thus
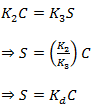 |
(38.24) (38.25) (38.26) |
This is the linear equilibrium sorption isotherm equation (38.3)
The reversible linear kinetic sorption model can also be written as,
(38.27) |
Where γ is a first order rate coefficient and is a constant.
The kinematic model discussed above considered that the rate of sorption has a linear relation with the concentration of the solute present . If we consider that the rate of sorption has a non-linear relation with the solute concentration, the reversible kinematic model becomes,
(38.28) |
Where K5 , K6 and K7 are the constant.
The advection-diffusion equation becomes,
(38.29) |
At the equilibrium condition ∂S/∂t = 0
Thus,
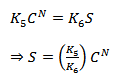 |
(38.30) (38.31) |
This is the Freundich equilibrium sorption isotherm.
The kinetic version of the Langmuir sorption model can be written as,
(38.32) |
Where is the maximum amount of solute that can be sorbed, K7 is the forward rate constant and K8 is the backward rate constant. This is known as bilinear adsorption model.
(38.33) |