(38.6) |
Here, R is the retardation factor. The advantage of linear sorption isotherm is that the equation (38.6) is apposite to solve mathematically. However, the linear sorption isotherm has two main drawbacks. The first one is that the relation between S and C is linear (which may not be the actual case) and there is an upper limit on S, i.e. there is an upper limit to the mass of solute that can be sorbed onto the solids.
Freundlich Sorption Isotherm
In 1909, Freundlich suggested a more general equilibrium isotherm. The non-linear Freundlich equilibrium isotherm (Fig. 38.2 a) is mathematically expressed as,
| S = KCN | (38.7) |
Taking on both sides, the relation becomes linear (Fig. 38.2 b)
logS = logK + NlogC |
(38.8) |
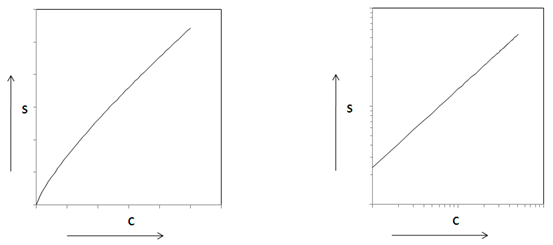 |
Fig. 38.2 Freundlich Sorption Isotherm (a) Normal scale (b) Log-Log scale |
The advection-diffusion equation can now be written as,
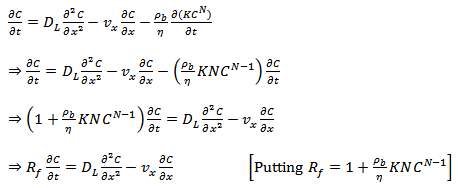 |
(38.10)
(38.13) |
Where, Rf is the retardation factor for a Freundlich sorption isotherm. The Freundlich equilibrium isotherm overcomes one of the main disadvantages of the linear equilibrium isotherm. However, couldn't overcome the second disadvantage that there is an upper limit to the mass of solute that can be sorbed onto the solids.
Langmuir Sorption Isotherm
In 1916, Irving Langmuir proposed a sorption isotherm which puts a limit on the maximum amount of solid that can be sorbed onto the solid matrix (Fig. 38.2 (a)).The Langmuir Sorption Isotherm can be expressed mathematically as,
(38.14) |
Where, α is a constant related to the binding energy between the solute and the soil solid (L/mg) and β is the maximum amount of solute that can be adsorbed by the soil solid (mg/Kg).