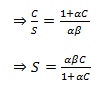 |
(38.15) (38.16) |
The advection-diffusion equation can now be written as,
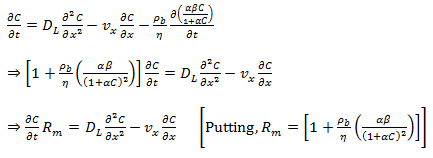 |
(38.17) (38.18)
(38.19) |
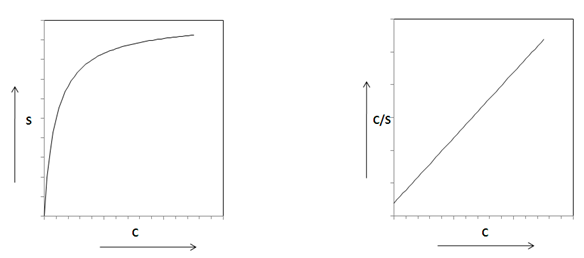 |
Fig. 38.2 Langmuir Sorption Isotherm (a) C - S plot normal scale (b) C - C/S plot in scale log - log scale |
Where, Rm is the retardation factor for the Langmuir sorption isotherm , α is an adsorption constant related to the binding energy (L/ mg) and β is the maximum amount of solute that can be absorbed by the solid (mg/kg).
The relation between C/S and C is linear in log - log scale as shown in Fig. 38.2 (b). This relationship can be used to obtain value of the constants α and β from experimental data .
Non-equilibrium Sorption Models
As discussed earlier, when the flow velocity is high, the sorb process may not reach the equilibrium state. In such a situation, kinematic model is more appropriate. The simple non equilibrium condition is the linear relation between the rate of sorption and the concentration of the solute remaining in solution. The relation can be mathematically represented as,
(38.20) |
Where, K1 is a first order decay rate constant.
The advection-diffusion equation becomes,
(38.21) |
In the above relation, it has been considered that the sorbed process is irreversible and therefore the solute cannot be desorbed once sorbed onto the soil solid. However, the sorbed process is a reversible one and the rate of sorption is also dependent to the amount of solid already sorbed onto the soil solid. Considering reversibility, the reversible linear kinetic sorption equation can be written as,
(38.22) |
Where, K2 is the forward rate constant and K3 is the backward rate constant.