Sorption Phenomenon
Sorption is a physical or chemical process by which the solute becomes attached with the soil solids. Sorption processes include adsorption, absorption, chemisorption and ion exchange. The molecules that are sorbed onto the solid are less mobile and they are also not available for phase transfer processes. As a result of the sorption processes, the movement of solute or mixing of solute becomes much slower than the movement of groundwater in an aquifer. Thus sorption induces a retardation effect to the movement of solute in groundwater aquifer.
As discussed earlier, the one dimensional advection dispersion equation can be written as
(38.1) |
After adding the sorption term, the equation becomes,
(38.2) |
Where, c is the concentration of the solute in liquid phase, DL is the longitudinal dispersion coefficient, vx is the average linear velocity of groundwater in x direction, ρb is the bulk density of the aquifer, s is the amount of solute sorbed per unit weight of the solid, η is the porosity of the aquifer and t is the time.
It has been observed from experimental studies that the capacity of a solid to remove a solute is a function of the concentration of the solute. The plot between the amount of solute sorbed onto the solid (S) and the concentration (C) of the solute is called Isotherm. When the sorb process is very fast compare to the flow velocity of water in the aquifer, the sorption process will reach an equilibrium state. This is described by an equilibrium sorption isotherm. On the other hand, when sorption process is very slow compare to the velocity of flow in the aquifer, it will not reach the equilibrium condition. In such a situation kinematic sorption model is necessary to model the sorption phenomenon. In the following section, we will discuss about the various isotherms that are used to model the equilibrium and kinematic sorption model.
Equilibrium sorption isotherm
Linear Sorption Isotherm
The linear sorption isotherm is defined as,
| S = KdC | (38.3) |
Where, S is the mass of solute sorbed per dry unit weight of solid (mg/Kg), C is the concentration of solute (mg/ L) and Kd is the distribution coefficient (L/kg). The equation (38.3) indicates that there is a direct relation between the amount of solute sorbed on to the solid and the concentration of the solute as shown in Fig. 38.1. The advection-diffusion equation can now be written as,
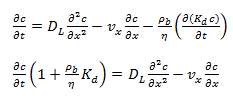 |
(38.4)
(38.5) |
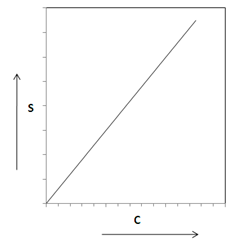 |
Fig. 38.1 Linear Sorption Isotherm |