The hydrazone derivatives of ketones undergo a rearrangement to form N-substituted amides in the presence of acidic reagents like H2SO4, PCl5, BF3 etc. This is known as Beckmann rearrangement (Scheme 13).
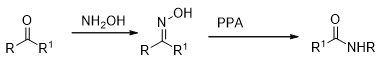
Scheme 13
The reaction is believed to proceed in the following manner. It is the group trans to hydroxyl group that always migrates under these conditions (Scheme 14).

Scheme 14
6.2.2.5 1.2-Addition Reaction
The reaction of carbonyl compounds with organometallic reagents is of unique importance in organic chemistry. These organometallic reagents are nucleophilic in nature and, thus, can attack on the carbonyl carbon. Though almost all classes of carbonyl compound react with aldehydes and ketones with varying results, in this section, only the reactions of aldehydes and ketones are discussed here.
Both aldehydes and ketones react with Grignard reagents to form an alcohol. Formaldehyde forms a primary alcohol as a result of this reaction while the rest of the aldehydes all form secondary alcohols. Similarly, the ketones form tertiary alcohols (Scheme 15).
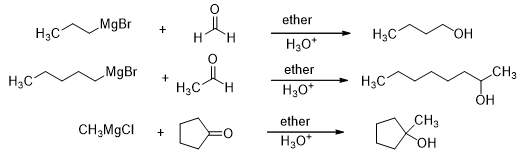
Scheme 15
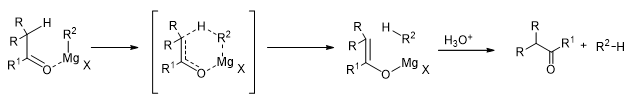
The actual composition of the Grignard reagents and the pathway of the reaction is a debatable issue, but it is clear that the Grignard reagents usually form associated structures among themselves (Scheme 16). This has been found to depend on the nature of the groups in the Grignard reagent as well as the solvent. It is believed that the reaction proceeds through a cyclic transition state involving the Grignard reagent, the carbonyl compound and the solvent molecules.
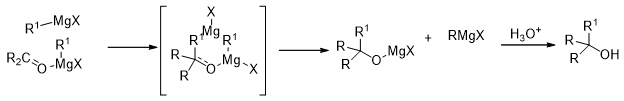
Scheme 16
However, when there are hydrogen atoms on the β-carbon of the Grignard reagent, then they tend to reduce the carbonyl compound, they themselves being oxidized to alkenes.
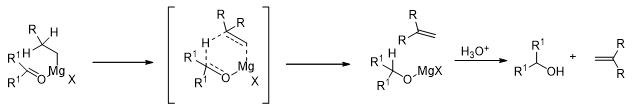
Similarly, sterically hindered ketones having hydrogen atoms on their α-carbons tend to be converted to enols, the Grignard reagent is reduced to form an alkane.
Another class of widely used organometallic reagents which act as a nucleophile are the organolithium reagents. The organolithium reagents are usually harder bases than the Grignard reagents and therefore show a greater reactivity.
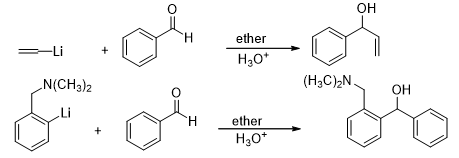
However, due to their higher reactivity, they are not prone to the side reactions occurring due to steric crowding as in Grignard reagents.