The Clemensen reduction discussed previously cannot be used acid sensitive carbonyl compounds. A method known as Wolff Kishner reduction may be employed in such circumstances (Scheme 8). The protocol involves the synthesis of the corresponding hydrazone from the ketone followed by reduction.
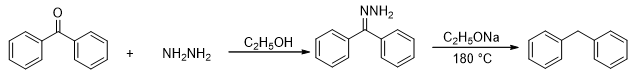
Scheme 8
The reaction is believed to proceed in the following manner (Scheme 9).
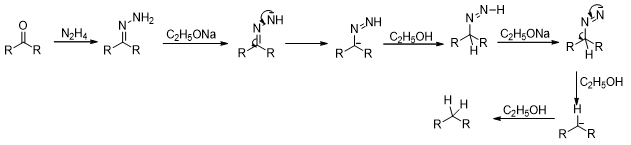
Scheme 9
Another type of reactions is the reaction of aldehydes and ketones with substituted amines (Scheme 10). Primary amines react with carbonyl compounds to give an imine (also called Schiff base). As shown above, the initial addition of the amine generates a carbinol amine which then undergoes β-elimination to give the imine as product. The reaction requires acid catalysis for the elimination step. However, the pH of the solution has to be maintained properly since a very low pH will result in protonation of the nucleophile amine.
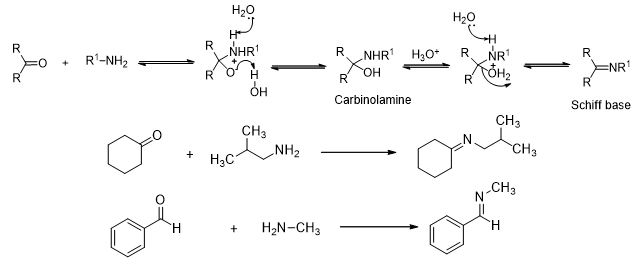
Scheme 10
Secondary amines may also participate in this reaction (Scheme 11). They add to aldehydes and ketones to form a carbinol amine which on dehydration leads to formation of an enamine. The pathway of the reaction is similar to the pathway shown above for the reaction of ketones with primary amines. The formation of enamine is of some synthetic importance as it is commonly used as an intermediate in many synthetic protocols.
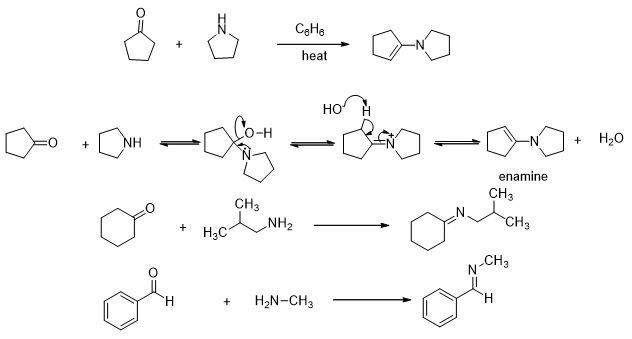
Scheme 11
One of the uses is in the selective alkylation as shown below. In this case, only the least hindered enamine is formed. This is because the enamine formation is thermodynamically controlled and, hence, the less substituted enamine is formed slowly. Thus, the alkylation takes place on that side only (Scheme 12).
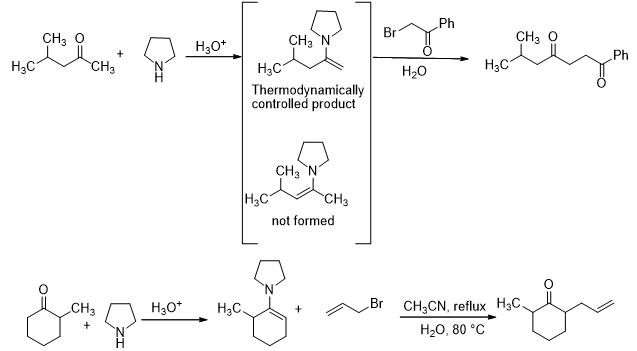
Scheme 12