6.2.2.3 Reduction
The reduction of aldehydes and ketones may be carried out by electrons generated from more electropositive elements under certain conditions (Scheme 1). Such electrons may act as a nucleophile and add to the carbonyl group to yield a radical anion often as ion pair with the metal cation. The blue colour generated by the aromatic ketones on reaction with sodium in the absence of air is an example of formation of such a radical.
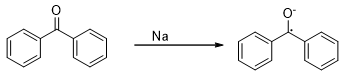
Scheme 1
The radical so formed can couple with a similar radical to form a 1,2-diol (pinacol) (Scheme 2). The reaction is however usually carried out using amalgamated magnesium.

Scheme 2
Another example of this phenomenon is the famed Clemensen reduction (Scheme 3). In this reaction, a ketone (usually) is treated with amalgated zinc and hydrochloric acid.
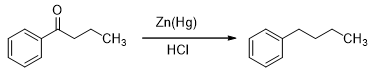
Scheme 3
Several pathways have been proposed for this reaction. However, the understanding of the mechanism of this reaction is still unclear. A probable mechanism is depicted in Scheme 4.
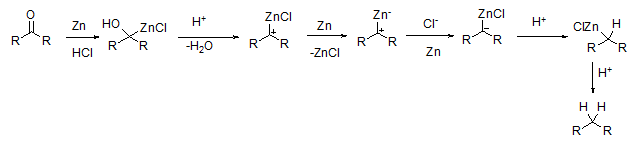
Scheme 4
Another application of this protocol is methylation of primary amines. The alkylation of amine often leads to polyalkylated amines. In case of methylation, the methylation can be stopped after one methyl group is added to the amine by treating it with formaldehyde to form an enamine followed by reduction (Scheme 5).
![]()
Scheme 5
6.2.2.4 Condensation
The aldehydes and ketones undergo another class of reactions which may be called addition elimination reaction or condensation reactions. In these reactions, they react with a reagent to form an adduct, which may then under suitable conditions undergo elimination of a water molecule to form the product. The most common reactions involve an attack by nucleophiles like hydroxylamine, phenyl hydrazine and its derivatives and semicarbazide and its derivatives. The general pathway for this reaction may be given as:
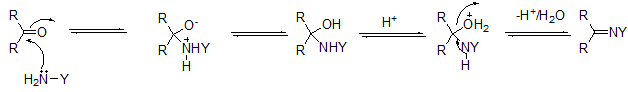
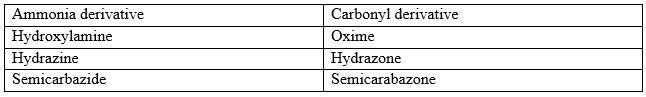
The strong nucleophiles such as hydroxylamine do not require assistance by any catalyst, however, weaker nucleophiles such as semicarbazone and its derivatives require general acid catalysis to activate the keto group. Thus, it may be expected that in basic and neutral solutions, the elimination of water is the rate limiting slow step which in an acidic solution, the attack of the nucleophile is the rate limiting step. The table below gives the names of the products of some common ammonia derivatives and ketones.
TThe most famous application of this category is the formation of 2,4-dinitrophenylhydrazone derivatives of ketones which are used to determine the presence of keto functionality (Scheme 6).
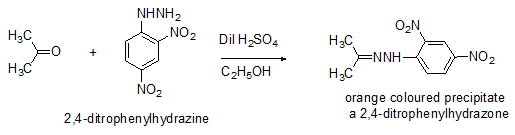
Scheme 6
Similarly, Girard’s reagent T and Girard’s reagent P are used in purification of carbonyl compounds (Scheme 7).
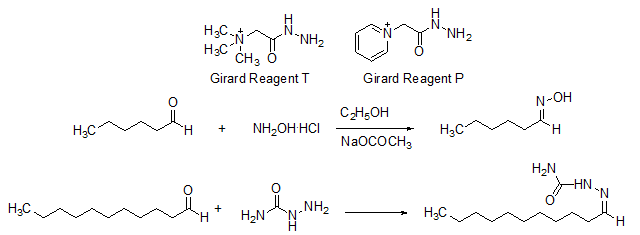
Scheme 7