Many electrons atom:
Schrödinger wave equation stood good for the hydrogen atom. This wave equation become highly complicated as the number of electrons in a system increased. To avoid all the difficulties and to have the equation a simpler form a several approximations have been considered. It has been found that the radial part of the wave function changes appreciably with change in Z . The angular part of the wave function remains nearly unchanged. Therefore, the basic shape of the orbitals that depends on angular part of the wave function, are same.
Effective nuclear charge and Slater´s rule:
As the nuclear charge and the number of electrons in an atom increases, n , l also increase. It has been shown earlier that with increase of n value the probability of finding of electron at r also increases. But it does not mean there is no electron density close to the nucleus. Electron density gradually decreases to the nucleus together with appearance of nodal plane. Nodal plane is the space where the probability of finding electron is zero and the number of nodal plane is governs by the formula, n - l -1.
Slater proposed that nuclear charge experienced by the electrons closer to the nucleus will be greater compared to the electrons far from the nucleus. This is because of the screening of nuclear charge by the inner electron core. This is called shielding effect. The screening or shielding efficiency decreases with decreasing to penetration properties of the orbitals. Hence, it follows the sequence,
s > p > d > f
Because of the screening effect the actual nuclear charge ( Z ) may be the replaced by Z * eff which is called effective nuclear charge. It is given by the equation,
Z * eff = Z - σ
σ is sum of the shielding contribution.
Estimation of σ is governs by the following rules,
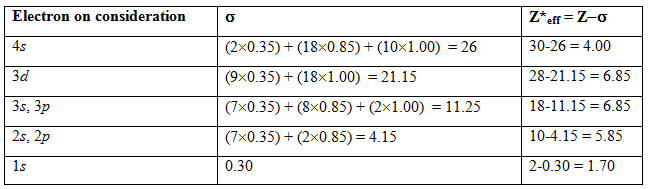
(i) The electrons are arranged in sequences of group with increase of n , and for equal n in order of l .
(1s ), (2s ,2p ), (3s ,3p ), (3d ), (4s ,4p ), (4d ), (4f ), (5s ,5p ), (5d ), (5f ),…….
(ii) The contribution of the electrons right to the electron that is under consideration is zero.
(iii) The contribution of each electron from same group 0.35, except 0.3 for 1s .
(iv) If the considered electron is under ( ns , np ) group, all the other electrons present at n-1 will contribute 0.85 each and all the other at n-2 or further left will contribute 1.00 each.
(v) If the considered electron is under ( nd ) or ( nf ) group, all the other electrons present at n (including ns , np ) or further left will contribute 1.00 each.
Rules are in tabulated form:
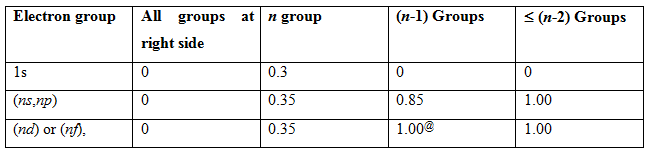 @ this value will be same for ns , np .
@ this value will be same for ns , np .