(D) Spin projection quantum number, m s :
An electron spins about its own axis. Because of this spinning an angular momentum is generated that is completely different from the orbital angular momentum. This angular momentum is expressed by spin projection quantum number ( m s ).
L z = m sh
For a given s value m s will have value in the range of +s to –s.
An electron has s = ½ spin, Hence, m s = +1/2, -1/2
Some other examples, s = 3/2, m s = +3/2, +1/2, -1/2, -3/2
s = 3, m s = +3/, +2, +1, 0, -1, -2, -3
This indicates, m s = 2 s + 1
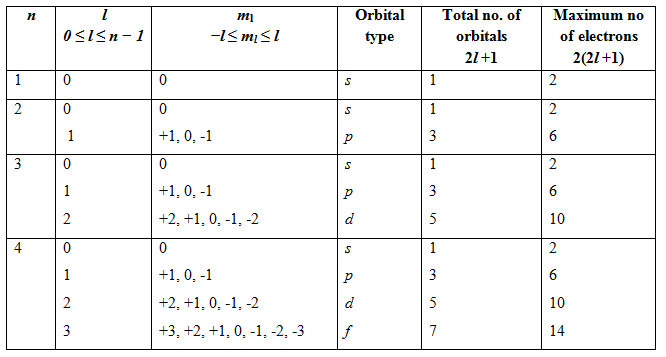
Table 1.3.
Sequence of energy level:
Rules:
- Orbital energies increases a ( n + l ) increases.
- If there are two orbitals with the same value of ( n + l ), the one with the smaller n has the lower energy.
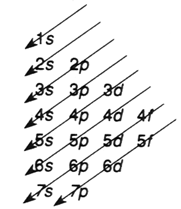
Figure 1.13. Energy levels sequence.