Quantum numbers:
Bohr-Sommerfeld aromic model and its later extensions, four quantum numbers ( n , l , m , s ) were introduced to explain the spectral features of atoms.
- The principle quantum number, n .
- The Azimuthal or angular momentum quantum number, l :
The Azimuthal or angular momentum quantum number determines the orbital angular momentum of an electron through the following relation. This specifies the shape of an atomic orbital, its bonding and bond angles.
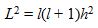
l may have any integral values between 0 to ( n -1). For l = 0, defines s orbital. l = 1, 2, 3, etc defines, p , d , f , etc respectively.
- The magnetic quantum number, m l :
This quantum number determines the energy level of an atom. The energy expressed as a function of n ,
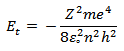
According to the above equation energy should decrease with increase of n value. Due to negative sign the energy also increases with increase in the value of n .
This quantum number determines the orientation of the angular momentum vector of an electron in the presence of external magnetic field along a specific axis (let consider along z axis).
L Z = m 1 h
For a given l value m 1 will be in between + l to – l .
i . e . m 1 = 2 l + 1
Example: for p orbital, l = 1, therefore, m 1 = +1, 0, -1. This indicates p orbital will have three different directions orientations in the presence of external magnetic field.
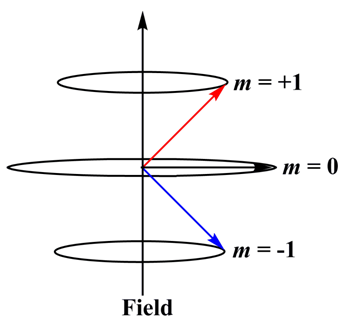
Figure 1.12. The orientation of orbital angular momentum vector in the presence of external magnetic field for l = 1.